Jaguar Health and Napo EU S.p.A have just announced that crofelemer, an investigative therapy for short bowel syndrome (SBS) has been granted Orphan Drug Designation by the EMA.
Thanks to this new designation, Napo EU is working on developing a clinical trial for SBS in both adults and pediatric patients.
Jaguar Health has an exclusive licensing agreement with Napo EU. This provides them royalties in Europe as well as a supply agreement.
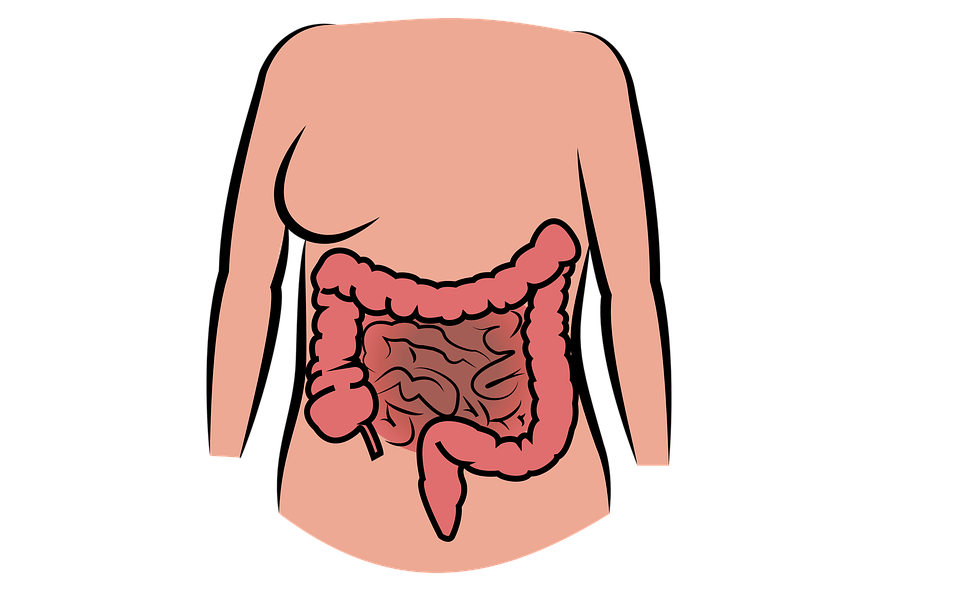
SBS
Short bowel syndrome is a rare condition caused by the malabsorption of nutrients and fluids. It is caused by congenital abnormalities, the surgical resection of the bowel, or a lack of absorption caused by another condition.
Patients who have lost a large portion of their intestines require IV fluids and nutrition daily. This leads to many daily challenges and often a decreased quality of life. It can be hard to attend work or school like normal or even to enjoy the simple pleasures of life.
Additionally, parenteral nutrition has its own risks. For instance, it can cause sepsis and even organ failure in extreme cases. Currently, the mortality rate for patients on parenteral nutrition for 5 years is 30%.
Crofelemer
The crofelemer treatment is a plant-based medication which is taken orally. It is derived from the sap of the bark of the Croton lechleri tree located in the Amazon.
Napo has created a sustainable program for harvesting this medication using fair trade practices. The company wants to ensure ecological integrity and quality.
Crofelemer has already been granted Orphan Drug Designation by the FDA for SBS.
You can read more about this treatment and its potential in SBS here.