NeuroVia, Inc. is a rare disease pharmaceutical company that primarily focuses on finding treatments to rare neurological diseases. Markedly, this private company is especially concerned about the effects of the rare X-linked adrenoleukodystrophy (try to say, or spell, that three times fast).
Funding for NeuroVia comes from Sanofi Genzyme and Novartis, a biotechnology company and a pharmaceutical company, respectively. Between these two companies, NeuroVia itself, and BioMed Ventures, as well as Enso Ventures, there is $14 million series A financing for NeuroVia’s ALD treatment candidate.
The treatment candidate, that is, which will target X-linked adrenoleukodystrophy.
Adrenoleukodystrophy, or, more conveniently, ALD, is a rare and deadly genetic disease. Since this is an X-linked disorder, it most commonly and severely affects males. The neurological disorder is characterized by a loss of the myelin sheath that encompasses neurons in the brain.
Because this crucial myelin sheath offers us the ability to think and control our movements, loss of it in adrenoleukodystrophy patients can be very problematic.
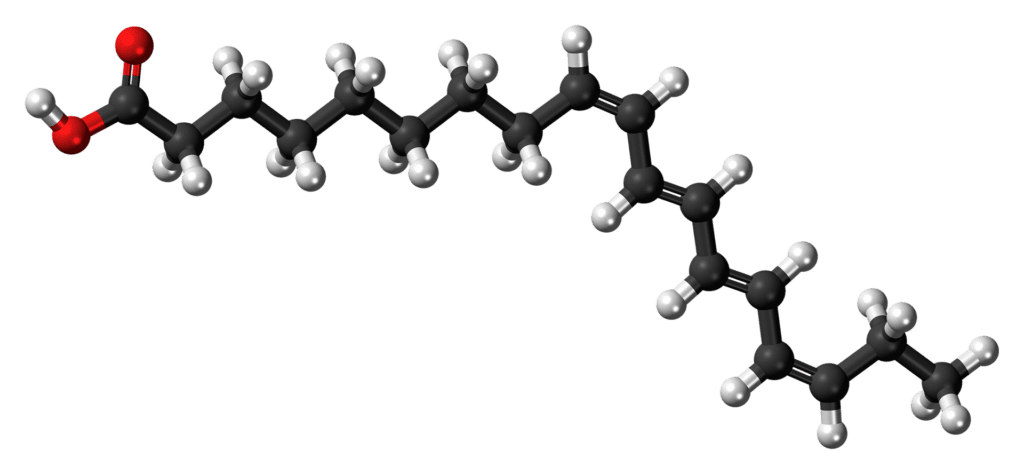
More scientifically speaking, X-linked ALD is defined by the build up of “very long chain fatty acids,” which eventually lead to this neurodegenerative disorder; the largest areas affected are the tissues in the brain, adrenal cortex and the spinal cord.
As a result of this disease, the central nervous system effects eventually produce two phenotypes: adrenomyeloneuropathy (AMN) and cerebral ALD (cALD). The former of these is followed by progressive motor problems, while cerebral ALD is associated with more severe neuroinflammation and eventually early death.
At the moment, there are just a couple of ALD treatments available, and even less are fully effective. Two notable ones are Lorenzo’s Oil and stem cell transplantation; Lorenzo’s Oil, a dietary supplement, is not significantly effective, and stem cell transplantation is known to have major limitations.
NeuroVia’s treatment, NV1205, is just a small molecule that proved in pre-clinical prototypes to regress the very long chain fatty acids (VLCFA) in the adrenal tissue, brain, and blood of mice that were lacking an adequately functioning ABCD1 gene.
The results from this make NeuroVia optimistic that this treatment could be a therapy that modifies the disease itself. They are hoping to develop a treatment for all X-ALD patient phenotypes.
Board member and co-founder of NeuroVia, Giovanni Ferrara, promised that he will keep the X-ALD community updated when the clinical trial sites become available and that the company is committed to allowing any qualified individual to participate in the studies.
Given how rare and how serious this condition is, I think we’re all looking forward to some big advancements from NeuroVia. To learn more about this project, read this.