Medical research and clinical trials are extremely important tools in learning more about specific diseases, as well as understanding and evaluating potential treatments. For example, the Phase 2 VIBRANT trial is working to evaluate vurolenatide for adults with short bowel syndrome (SBS). According to a news release from clinical stage biopharmaceutical company 9 Meters Biopharma, Inc. (“9 Meters”), positive preliminary data from the study is now available.
Evaluating Vurolenatide
To begin, let’s take a look at exactly what vurolenatide is. Vurolenatide is an injectable GLP-1 receptor agonist which has an extended half-life. According to 9 Meters, vurolenatide is:
designed specifically to slow digestive transit, thereby mitigating the SBS hallmark of excessive gastric motility due to deficient GLP-1 in the shortened intestine. This mechanism is intended to improve intestinal absorption of nutrients and water and reduce diarrhea for all patients with any type of post-surgical SBS, regardless of their parenteral support requirements.
Additionally, patients only need to be dosed around 2x monthly, making it a more efficient and less intrusive therapeutic option.
In a prior Phase 1b/2a study, researchers found that vurolenatide was relatively safe and well-tolerated. The Phase 2 VIBRANT study shows similar findings. Patients received either 50mg vurolenatide weekly or bi-weekly, 100mg bi-weekly, or a placebo. The preliminary findings, which include data from 11 adults with SBS, show that the treatment is safe, effective, and well-tolerated. Three participants (27%) were able to reduce their need for parenteral support. Seven participants (63.6%) also saw improved total stool output, reducing the volume by 10-25%.
Adverse reactions included nausea and vomiting. Due to the relatively positive results, 9 Meters hopes to pursue a Phase 3 study in the future – possibly launching the study by the end of 2022.
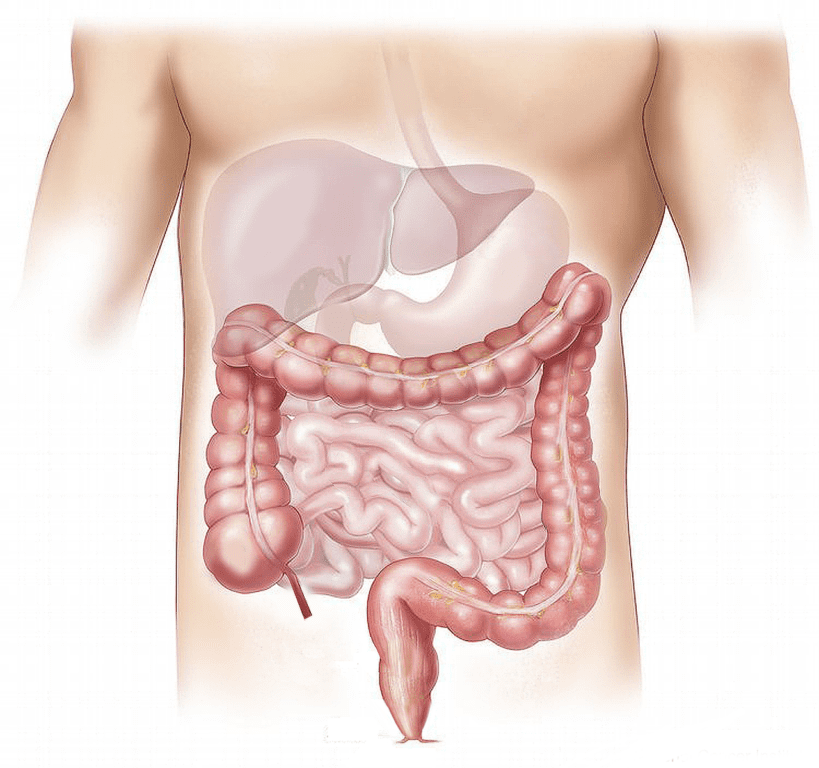
An Overview of Short Bowel Syndrome (SBS)
Short bowel syndrome (SBS) is a disorder which occurs either due to partial or total removal of the small intestine due to injury, disease, poor motility, or birth defect. For example, diseases causing loss of small intestine function, radiation or chemotherapy damage, Crohn’s disease, and cancer may all cause SBS. As a result, patients are unable to effectively absorb nutrients and water. SBS is a variable condition, meaning patients may vary in experience and symptom severity. Potential symptoms of SBS can include:
- Fatigue
- Persistent diarrhea
- Abdominal cramping and bloating
- Heartburn
- Anemia (low red blood cell count)
- Unintended weight loss
- Gallstones and kidney stones
- Fatty liver
- Nausea and vomiting
- Excessive gassiness
- Dehydration
- Easy bruising
- Bone pain
- Food intolerances