The Loma Linda University Eye Institute in California performed its first-ever procedure using a FDA-approved procedure, which can deter the effects of keratoconus – a rare progressive disease of the cornea, which you can read more about here.
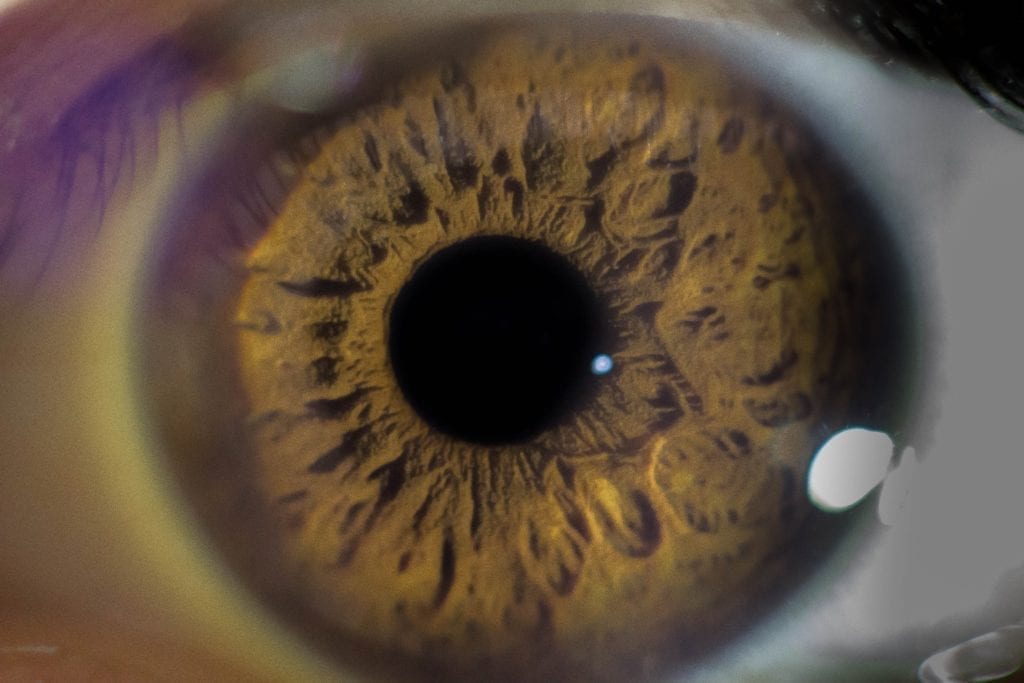
Keratoconus is characterized by progressive thinning of the cornea, often resulting in a conical bulge toward the center of the eye. Symptoms include blurred vision, increased sensitivity to light and sudden worsening or clouding of vision.
The procedure is called corneal collagen cross-linking – and it combines the use of UV light with riboflavin (Vitamin B-2) eye drops. This combination develops support across the cornea, hardening the surface to resist bulging – called corneal ectasia. This bulging causes deterioration of eyesight.
While this damage to eyesight is not reversible, the treatment goal is to stop further deterioration and can prevent the need for a corneal transplant.
The procedure takes about an hour per eye – with patients typically being able to go home on the same day.
Click here to read about the very first patient to receive this new treatment!