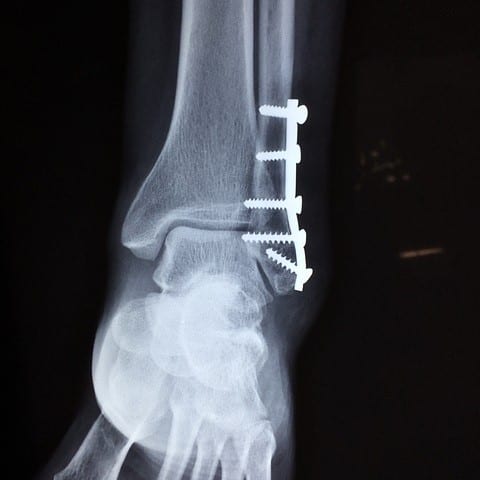
Osteoporosis is known to affect over two hundred million people worldwide. According to a recent article in the Endocrinology Advisor, fractures of the spine or hip due to reduced bone mineral density are the primary cause of the disability and death of older women.
But for the past two decades, doctors in the U.S. have seen a reduction in hip fractures. Some credit can be given to improved nutrition, an increase in physical activity, and guidance about preventing falls.
However, these improvements also coincide with the 1995 introduction of a medication that increases bone density known as bisphosphonates. This class of drugs is known to be effective in the prevention or mitigation of osteoporosis which can cause bone fractures, pain, or hospitalization.
FDA Issues A Drug Safety Update
In February 2018, the FDA issued an update to its March 2010 Drug Safety Review of bisphosphonates and femur fractures.
Its review included information regarding the risk of uncommon fractures of the thigh (femur) in connection with bisphosphonates. The information was added to warning labels on all bisphosphonate drugs that were FDA approved for the treatment of osteoporosis.
The most common BPs are:
- Alendronate (Fosamax) (see “Lawsuit”)
- Risedronate (Actonel)
- Ibandronate (Boniva)
- Zoledronic acid (Reclast)
It has not been determined if bisphosphonates cause these unusual fractures but they have mostly been reported in patients using bisphosphonates.
Reducing the Risk of Bisphosphonates
One consideration to reduce adverse events during long term bisphosphonates treatment may be interrupting administration of the drug and having periodic reassessments. The researchers felt a need for further evaluation of this concept.
A study of 1,973 patients, (ninety-six percent were women), observed the risk of fractures during a BP “holiday.” This was compared to the uninterrupted use of BP.
Researchers analyzed patients with a four year or greater history of taking BP. The baseline risk of fractures was set according to data submitted by referring clinicians. Additional information was obtained intermittently over a twenty-four-month period after the onset of the study.
Three months after the onset of the study, 1250 subjects were continuing BP treatment. During that same period, six hundred patients had interrupted their treatment.
At the six-month interval, 884 people reported that they had interrupted BP treatment. However, the median time for the overall “holiday” varied from one to two years by the end of the study.
Summary
In summary, study results show that although risks for vertebral fractures were somewhat lower in patients taking the BP holiday, the difference was not significant. Only in the presence of a prevalent vertebral fracture was a long BP holiday associated with an increased risk.