According to a story from PR Newswire, new international treatment recommendation guidelines have recently been updated for nontuberculous mycobacterial (NTM) lung disease. The update pertains to a variant known as Mycobacterium avium complex (MAC) lung disease. This update recommends the use of amikacin liposome inhalation suspension (marketed as ARIKAYCE®) as a treatment for patients with MAC lung disease as part of a antibacterial regimen in patients who have failed to see their symptoms resolve after six months of treatment. ARIKAYCE was developed by biopharmaceutical company Insmed Inc.
About MAC Lung Disease
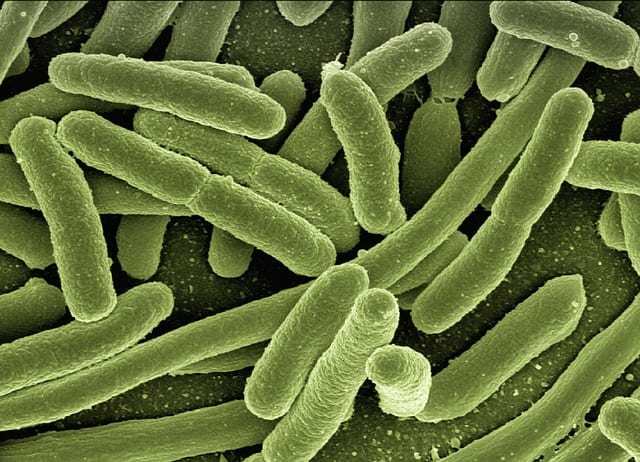
MAC lung disease is a type of rare, infectious lung disease. It is caused by one of two species of bacteria: Mycobacterium avium or M. intracellulare. These widespread bacteria are almost never a threat to humans under normal circumstances; however, in patients with weakened immune systems, such as those living with HIV/AIDs, undergoing chemotherapy, or living with primary immundeficiency, these bacteria present a risk. Symptoms of infection include cough, fever, night sweats, weight loss, sputum production, lethargy, diarrhea, and infection in other areas of the body. The treatment of MAC lung disease often involves the use of a combination of two or three antibacterial drugs. It can take some time for the infection to be eradicated, and treatment may last for a year or more. Outcomes can vary and often depend on the underlying condition of the patient. To learn more about MAC lung disease, click here.
A Long Awaited Update
These guidelines are used worldwide for the treatment of the disease as well as other forms of NTM lung disease. It has been more than ten years since these guidelines were last updated, and this update marks a significant advancement in the treatment of this lung disease.
About ARIKAYCE
ARIKAYCE is the only medication approved by the US Food and Drug Administration (FDA) to treat this lung disease. This approval followed results from a phase 3 trial in which the medication, when used in combination with a background therapeutic regimen, was able to eliminate evidence of infection in 29 percent of patients after six months of treatment compared to just 9 percent in patients using solely the background regimen.