Takeda Pharmaceutical Company has announced breakthrough results from Phase 3 trials of zasocitinib (TAK-279), an innovative oral treatment for moderate-to-severe plaque psoriasis that offers patients a promising new option for achieving clear skin. The positive findings reported by Drugs,com, represent a significant advancement in psoriasis treatment, potentially ushering in a new era of convenient oral therapies that address this chronic inflammatory condition affecting millions worldwide.
Strong Clinical Efficacy
The two pivotal Phase 3 studies demonstrated remarkable efficacy across multiple measures. Zasocitinib achieved superiority over placebo on both co-primary endpoints, static Physician Global Assessment (sPGA) 0/1 and Psoriasis Area and Severity Index (PASI) 75, at week 16. Notably, patients showed measurable improvement as early as week 4, with response rates continuing to climb through week 24.
The results were particularly impressive for near complete and complete skin clearance. More than half of patients treated with zasocitinib achieved PASI 90 (clear or almost clear skin), while approximately 30 percent achieved PASI 100 (completely clear skin) at week 16. The studies met all 44 ranked secondary endpoints, including favorable comparisons against both placebo and apremilast, a commonly used oral treatment.
Mechanism of Action
Zasocitinib works by selectively inhibiting TYK2 (tyrosine kinase 2), an enzyme that plays a central role in immune-mediated inflammation. What sets this drug apart is its exceptional selectivity, it demonstrates more than 1-million-fold greater selectivity for TYK2 compared to other JAK enzymes. This precision targeting means the drug can maintain 24-hour inhibition of IL-23 and other disease-driving immune pathways while minimizing effects on JAK1, 2, and 3, which regulate broader biological processes. This targeted approach may reduce the risk profile associated with broader JAK inhibition.
Safety Profile
Safety data from the Phase 3 trials was reassuring. Zasocitinib was generally well-tolerated, with the most common adverse events being upper respiratory tract infection, nasopharyngitis, and acne—consistent with findings from earlier Phase 2b studies. Importantly, no new safety signals were identified through week 24, suggesting the drug maintains a favorable safety profile even with extended use.
Impact on Patient Lives
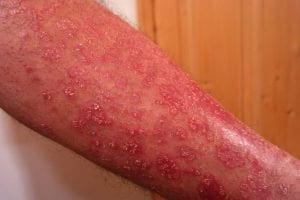
For the estimated 64 million people living with psoriasis worldwide, approximately 80-90 percent having the plaque form, these results offer hope. Plaque psoriasis causes significant patient burden beyond visible skin symptoms. The raised, red, scaly patches cause intense itching and burning that disrupts daily life, while the visible nature of the condition often leads to social isolation and mental health challenges. A highly effective once-daily oral medication could substantially improve quality of life for these patients.
Path Forward
Takeda plans to present detailed results at upcoming medical conferences and intends to submit a New Drug Application (NDA) to the U.S. Food and Drug Administration in fiscal year 2026. Zasocitinib is also being evaluated in additional studies for psoriatic arthritis, Crohn’s disease, ulcerative colitis, and other immune-mediated inflammatory conditions, suggesting the drug’s potential extends beyond psoriasis treatment.