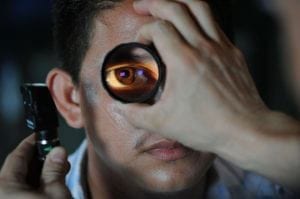
In the past, genetics were hypothesized to be a potential cause of glaucoma and eye disease. Now, British researchers definitively linked SPATA13 gene mutations with a form of glaucoma called primary angle-closure glaucoma (PACG). With an estimated 50% of glaucoma-related blindness caused by PACG, understanding the genetic cause can improve detection and treatment in the future. You can find the full research article in PLOS Genetics.
Glaucoma
Glaucoma refers to a group of conditions that result in progressive optic nerve damage. The liquid in the front of the eye doesn’t circulate properly through a mesh-like channel (called trabecular meshwork). Instead, that channel gets blocked. The liquid builds up, increasing in pressure and harming the optic nerve. Because the optic nerve helps transmit images to the brain, damage can cause partial or complete vision loss.
There are multiple rare forms of glaucoma, such as congenital glaucoma, pseudoexfoliation syndrome, neovascular glaucoma, and irido corneal endothelial syndrome (ICE). However, there are more common forms as well, such as PACG. Symptoms vary but can include vision loss, headache, blurred vision, eye pain, and halos around lights. Learn more about glaucoma here.
Primary Angle-Closure Glaucoma (PACG)
According to the International Glaucoma Association, PACG occurs when the iris prevents the liquid from draining from the eye through the trabecular meshwork. PACG can also refer to angle closure glaucoma and chronic angle closure glaucoma. The IGA lists risk factors for PACG as:
- Ethnicity. PACG is more common in Asian individuals or those in Eastern countries.
- Age. Most cases occur after age 40.
- Sex. Females experience angle closures in the eyes at a higher rate than males.
- Family history. If someone in your family has glaucoma, you are more at risk of developing PACG.
Symptom onset is usually fairly quick in acute cases. However, those with chronic angle closure may never have symptoms. For those that do, symptoms of PACG include:
- A rapid rise in eye pressure with associated pain
- Nausea and vomiting
- A red eye
- Cloudy or blurry vision
- Halos around light sources
Some individuals also experience “attacks” in the evening, which contributes to cloudy vision and eye discomfort. PACG symptoms can be reduced or reversed with early treatment.
Learn more about PACG here.
Discovering the New Mutation
Glaucoma-related blindness impacts an estimated 80 million people worldwide, with over half of the cases attributed to PACG. Researchers wanted to explore the underlying genetic causes through familial analysis.
A family study began at Moorfields Eye Hospital in London, England. Some family information includes:
- The family is Caucasian.
- They lived in London for 7 generations, starting in 1812.
- Glaucoma runs in the family.
- The initial member of the study (proband) had 9 aunts and uncles.
- The proband’s mother got surgery for PACG.
- The proband was treated for ocular hypertension, or high eye pressure, from ages 52 to 67.
- The initial member, her sister, and her daughter had the same plateau iris configuration. This means the angle of the iris pressured up against the trabecular meshwork.
- Eventually, researchers managed to contact 63 family members. Of these, 12 (19%) either had primary angle-closure disease, suspected PACG (called primary angle-closure suspect, or PACS), or PACG.
Analyzing the family’s genes found various SPATA13 gene mutations. All those in the 7th generation shared a common SPATA13 mutation. However, in other generational levels, those with PACG also had other mutations of the same gene. Within the eye, SPATA13 was being expressed in damaged areas. This affirmed that SPATA13 mutations played a role in the development of eye disease.
Intriguingly, this is the first evidence and first study to identify SPATA13 mutations as the genetic reasoning behind PACG. According to Dr. Ahmad Waseem, this will help improve detection and treatment moving forward through novel and targeted treatments and screening processes.
Read the original article here.