In a news release from May 1, 2021, Chiesi Global Rare Diseases, a business unit of the pharmaceutical company the Chiesi Group, shared its FDA acceptance of FERRIPROX (deferiprone). Altogether, the therapy is designed to treat iron overload in patients with sickle cell disease (SCD) or other forms of anemia or thalassemia. The FDA approved the treatment for patients 3+ years old using the oral solution, or 8+ years old using the tablets.
FERRIPROX
So what is FERRIPROX? This synthetic iron chelator helps to reduce iron concentration in cells. Once it enters cells by penetrating cell membranes, FERRIPROX works to clear toxic iron from tissues and fluids. Typically, FERRIPROX is administered in 1000mg doses 2x daily. Ultimately, Chiesi worked to develop this treatment to assist patients with renal issues associated with SCD, for whom treatment options are not always available.
Data from clinical trials suggest that FERRIPROX benefits patients with SCD. Some findings include:
- FERRIPROX confers sustained benefits, including progressively decreasing liver iron concentrations. For example, after 3 years of treatment, FERRIPROX reduced liver iron concentration from a mean of 14.93 mg/g dw to 10.45 mg/g dw.
- Although FERRIPROX was relatively safe and well-tolerated, some adverse reactions did occur. These include:
- Headache
- Nausea and vomiting
- Abdominal, back, joint, and bone pain
- Fever
- Cough
- ALT and AST increases
- Low neutrophil (white blood cell) count
If you are taking FERRIPROX, you may notice dark or discolored urine. This is normal and not a cause for concern. If you are interested in learning more about prescription information, refills, or financial assistance, please call 866-758-7071 and ask about the Chiesi Total Care Program.
Sickle Cell Disease (SCD)
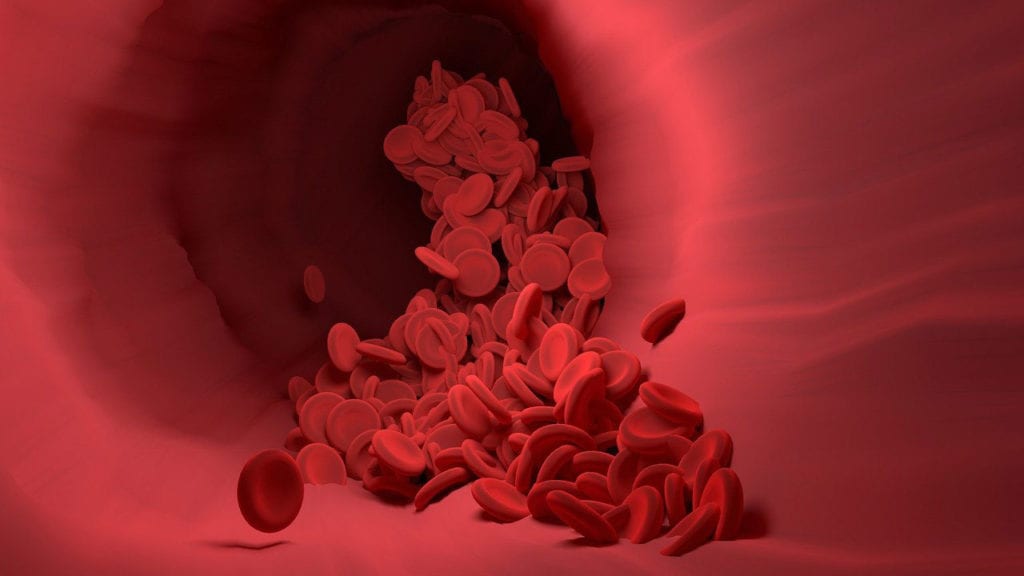
Overall, sickle cell disease (SCD) consists of a group of hematological disorders characterized by sickle-shaped red blood cells (RBCs). Normally, RBCs are spherical, allowing for easy passage throughout the body. But in SCD, malformed RBCs get caught on the walls of blood vessels, causing blockages and poor blood flow. Gene mutations, specifically those in hemoglobin-producing genes, cause SCD. Because the condition is inherited in an autosomal recessive pattern, patients must inherit one defective gene from each parent to develop SCD. Sickle cell anemia is the most common, and most severe, form of SCD. In most cases, SCD is diagnosed in early childhood.
Symptoms include:
- Swelling of the hands and feet
- Jaundice (yellowing of the skin and eyes)
- Chronic pain
- Gallstones
- Pulmonary hypertension (PH)
- Frequent or recurrent infections
- Fatigue
- Delayed growth and development
- Retinal detachment and/or vision loss
- Pain crisis
- Organ damage
- Note: Organ damage caused by SCD typically occurs in the bones, kidneys and liver, spleen, eyes, brain, lungs, heart, skin, or joints.