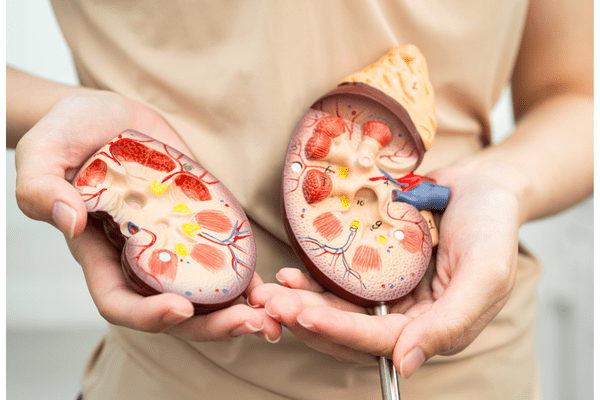
Merck & Co. announced positive results from two Phase 3 studies of its kidney cancer drug Welireg, signaling potential expansion into earlier stages of disease. As reported on BioPharmaDive, in one trial, Welireg combined with Merck’s immunotherapy Keytruda after surgical tumor removal significantly extended disease-free survival compared to Keytruda alone. Another study found that pairing Welireg with Eisai’s Lenvima delayed disease progression more effectively than Exelixis’ Cabometyx in patients whose cancer returned after prior Keytruda treatment.
Welireg, an oral HIF-2 alpha inhibitor first approved in 2021 for rare tumors, is currently indicated for advanced kidney cancer following immunotherapy and targeted therapy. Merck projects that success in the adjuvant setting could dramatically increase revenue, with analysts estimating potential sales exceeding $6 billion. The drug’s high monthly cost and extended treatment duration further underscore its commercial impact.
While both trials demonstrated meaningful improvements in progression metrics, overall survival data remain pending. Regulatory review will likely consider efficacy alongside safety, particularly given Lenvima’s side effect profile. Merck’s progress positions Welireg as a key growth driver as Keytruda approaches patent expiration in 2028, amid competition from similar agents like Arcus Biosciences’ casdatifan.