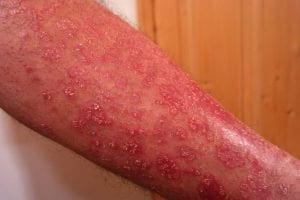
FibroBiologics Inc, a clinical-stage biotechnology company, has announced a significant breakthrough in regenerative medicine by submitting a Phase 1/2 Investigational New Drug (IND) application to the US Food and Drug Administration for CYPS317, a groundbreaking allogeneic fibroblast spheroid-based therapy designed to treat moderate to severe psoriasis. According to PharmaBiz.com, this submission represents a pivotal transition from preclinical research to human clinical trials and underscores the company’s commitment to developing novel therapeutics for chronic inflammatory diseases.
CYPS317 is built on FibroBiologics’ proprietary fibroblast spheroid platform, leveraging the company’s extensive intellectual property portfolio of over 270 issued and pending patents. The therapeutic approach differs fundamentally from existing treatments by utilizing engineered fibroblast cell spheroids to target the underlying mechanisms of psoriasis rather than simply managing surface symptoms.
The IND submission is supported by compelling preclinical data that demonstrates CYPS317’s remarkable efficacy and durability. In animal models, a single dose of the therapy matched or exceeded the therapeutic effects of multiple doses of anti-IL-23 monoclonal antibodies, currently among the most advanced psoriasis treatments available. Notably, CYPS317 also produced significant reductions in disease recurrence, suggesting that this approach may offer more durable benefits compared to conventional biologics. This distinction is particularly important for patients seeking long-lasting symptom control with reduced treatment burdens.
The therapy’s multi-faceted mechanism of action combines extracellular matrix signaling with both localized and systemic immunomodulation, potentially restoring normal tissue homeostasis in psoriasis patients. This comprehensive biological activity addresses the complex nature of the disease, which involves immune system dysregulation, inflammatory cascades, and skin tissue dysfunction.
Psoriasis remains a significant unmet medical need despite recent biologic advances. The disease affects over eight million adults in the United States alone, often severely impacting quality of life, productivity, and mental health. While newer biologic treatments have improved outcomes, many patients continue to struggle with inadequate responses, frequent relapses, and safety concerns, highlighting the urgent need for alternative therapeutic approaches.
Pete O’Heeron, founder and CEO of FibroBiologics, emphasized the strategic importance of this milestone, stating that CYPS317’s IND filing advances the company’s goal of achieving clearance for all four product candidates by the end of 2026. He highlighted the potential of fibroblast spheroid therapeutics to “redefine how chronic inflammatory disorders are treated” while improving disease outcomes and patient experiences.
Chief Scientific Officer Hamid Khoja underscored the innovative nature of the approach, noting that preclinical evidence suggests CYPS317 harnesses unique biological activity that could transform treatment paradigms for chronic inflammatory conditions beyond psoriasis.
The IND application includes comprehensive preclinical pharmacology, safety, and manufacturing data demonstrating the therapy’s mechanism of action, durability of effect, and safety profile. With FDA review underway, FibroBiologics is positioned to launch first-in-human trials, potentially offering psoriasis patients a revolutionary treatment option that combines durability with improved safety profiles. This advancement exemplifies the growing promise of cell therapy and tissue regeneration in addressing previously intractable chronic diseases.