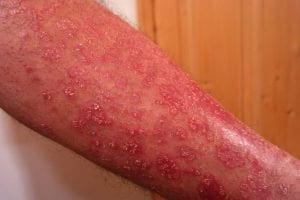
Johnson & Johnson announced promising long-term efficacy and safety data for Icotyde (icotrokinra), the first and only targeted oral peptide that precisely blocks the IL-23 receptor for plaque psoriasis treatment. As reported by Drugs.com, one-year results from Phase 3 trials presented at the 2026 American Academy of Dermatology Annual Meeting demonstrate sustained skin clearance and a reassuring safety profile, positioning this once-daily oral therapy as a meaningful advancement in systemic psoriasis treatment.
Sustained Efficacy Through One Year
The ICONIC-ADVANCE 1 and 2 trials revealed impressive durability of Icotyde’s therapeutic benefits. In ADVANCE 1, complete skin clearance (PASI 100) rates increased from 41% at Week 24 to 49% at Week 52. Similarly, ADVANCE 2 demonstrated improvement from 33% to 48% over the same period, indicating that continued treatment yielded incremental skin clearance benefits for patients.
Notably, patients who started Icotyde treatment later—switching from placebo to active treatment at Week 16—achieved comparable skin clearance rates by Week 52 (50% and 43% in ADVANCE 1 and 2, respectively) as those receiving the full 52-week treatment course. This finding suggests that delayed therapy initiation does not compromise long-term outcomes, an important consideration for treatment sequencing decisions.
Adolescent Population Shows Exceptional Response
The ICONIC-LEAD trial specifically evaluated adolescents 12 years and older, a population previously limited in oral systemic psoriasis therapy options. Nearly 60% of adolescents achieved completely clear skin at Week 52, with 86% achieving PASI 90 response at one year and 92% maintaining that improvement from Week 24 through Week 52. This performance represents a transformative treatment option for younger patients managing this chronic disease.
Dr. Jennifer Soung, Director of Clinical Research at Southern California Dermatology, highlighted the clinical significance: “For the first time, patients 12 and older have access to a novel therapy capable of delivering sustained skin clearance and a favorable safety profile in a once-daily pill.”
Consistent Safety Profile
Beyond efficacy, Icotyde demonstrated a remarkably consistent and favorable safety profile through the full year of treatment. The adverse event profile observed through Week 52 remained consistent with that observed at earlier timepoints (Weeks 16 and 24), with no new safety signals identified despite extended exposure. Importantly, overall adverse event and infection rates with Icotyde were lower than observed with deucravacitinib through Week 24, a comparative finding that strengthens its safety profile relative to competing therapies.
Mechanism and Clinical Significance
Icotyde’s unique mechanism, precise blocking of the IL-23 receptor, addresses the inflammatory cascade underlying plaque psoriasis pathogenesis. Unlike broader JAK inhibitors, this targeted peptide approach provides specific inhibition of IL-23 signaling, potentially reducing off-target effects associated with less selective mechanisms.
Expanded Treatment Access
The one-year data underscore Icotyde’s approval for adults and pediatric patients 12 years and older weighing at least 40 kg. The convenient once-daily oral administration—taken with water 30 minutes before eating—represents a significant quality-of-life advantage compared to injectable or infusion-based therapies.
These sustained, long-term results position Icotyde as a first-line systemic therapy capable of addressing significant treatment gaps in moderate-to-severe plaque psoriasis across diverse age groups, with clinical efficacy and safety characteristics that advance the therapeutic landscape for this chronic inflammatory condition affecting millions globally.