A New Shield for the Blood Supply: Grifols Earns FDA Approval for Malaria Test
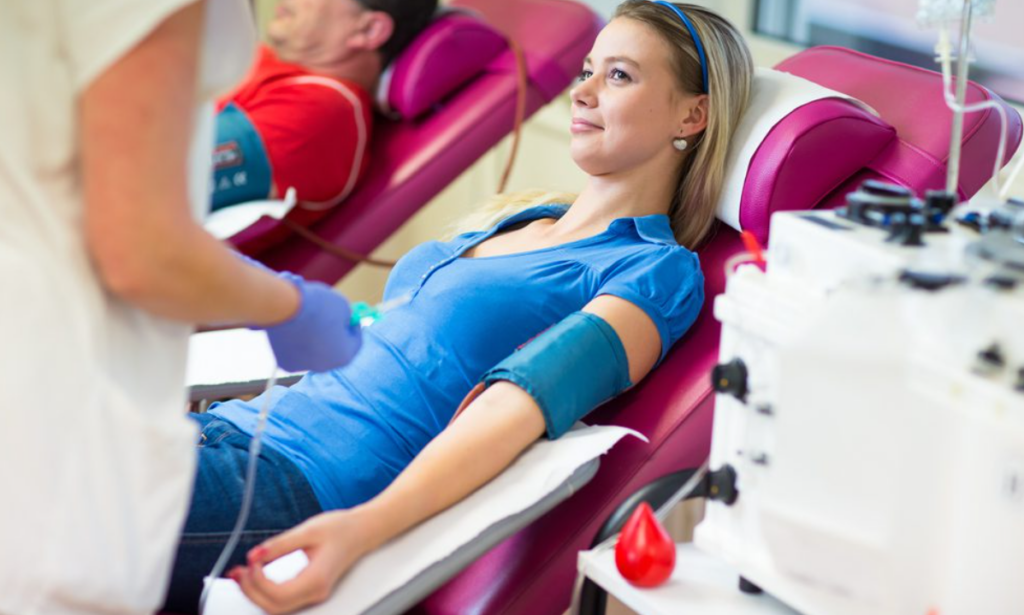
Grifols has reached a significant milestone in transfusion medicine with the FDA approval of its Procleix Plasmodium Assay. This advanced screening tool, which has been in use in Europe since 2022, is now set to modernize how the United States protects its blood supply from malaria.
Moving Beyond the Questionnaire
For years, blood banks have relied largely on “travel deferrals”—asking donors if they’ve recently visited malaria-endemic regions. This protocol for keeping the parasite out relies on self-reporting and it often sidelines healthy donors based on their passport stamps rather than their actual health.
The Procleix Plasmodium Assay changes the game by using Nucleic Acid Testing (NAT) to look for the parasite itself. By detecting the ribosomal RNA of the five major Plasmodium species, the test can identify infections even in “silent” carriers who show no symptoms.
Key Features of the Assay
- High Sensitivity: Detects even minute levels of the parasite by targeting abundant ribosomal RNA.
- Broad Spectrum: Covers all five human-infecting species: P. falciparum, P. knowlesi, P. malariae, P. ovale, and P. vivax.
- Automation-Ready: The assay runs on the Procleix Panther System, a fully automated platform designed for high-volume labs that need results without the manual heavy lifting.
Why This Matters
Malaria remains a global titan of disease, responsible for over 600,000 deaths annually. In the context of blood transfusions, even a tiny amount of contaminated blood can be fatal to a vulnerable recipient. Travel to malaria prone areas has been the number one cause of travel deferrals at blood banks, with the number deferred estimated to be 150,000 to 160,000 each year. In addition, these numbers don’t count the people who look at the donation requirements online, believe they may have travelled to a Malaria endemic area, and do not even show up to donate. These people may be two or three times higher than those actually deferred.
By integrating this test into standard U.S. screening, blood centers can achieve a “double win”:
- Enhanced Safety: A more rigorous barrier against transfusion-transmitted malaria.
- Increased Availability: The potential to reduce the number of deferred donors, keeping more life-saving blood on the shelves. The blood supply in the USA has reached critically low levels several times in the past few years. “The Procleix Plasmodium Assay enhances the ability of blood banks in the United States to provide safer blood transfusions,” noted Antonio Martínez, President of Grifols Diagnostic.
With this approval, Grifols has helped ensure that the act of giving blood remains as safe for the receiver as it is noble for the donor and may help to increase the blood supply for all. That, is indeed, Patient Worthy. Let’s hope that the American Red Cross, OneBlood and all other blood banks begin utilizing this system immediately.


-300x300.jpg)



