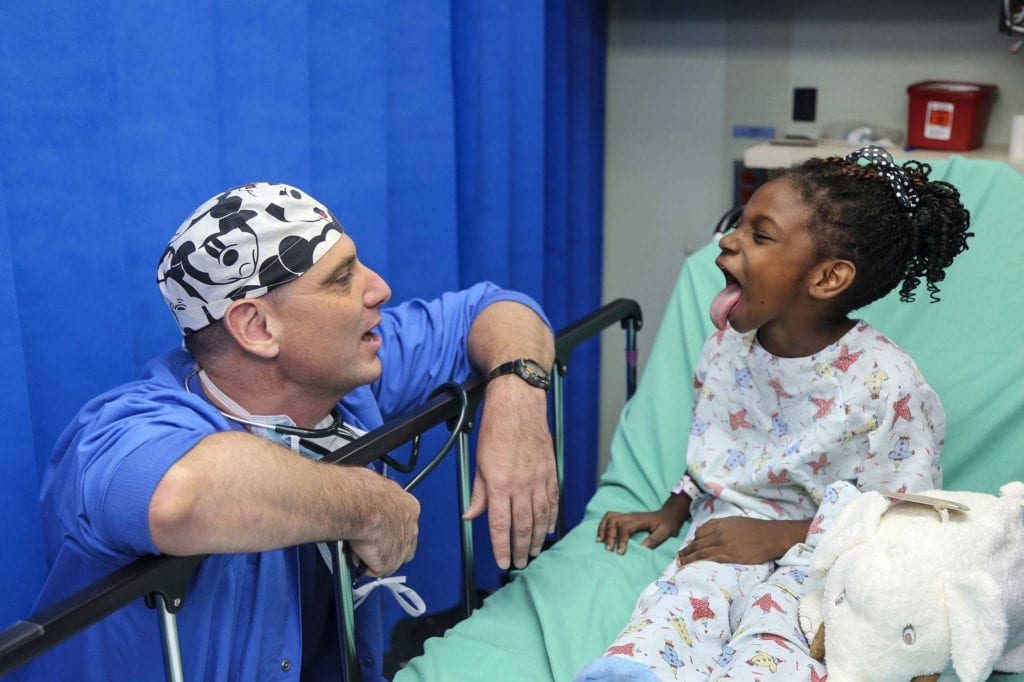
The Importance of Emotional Support for Children with Chronic Illnesses
It wasn’t until the 1980s that the medical field really started talking about the need to emotionally support patients, especially those with chronic illnesses. This is partly due to the…