Initially, belimumab (sold under the brand name BENLYSTA) was approved for the treatment of patients for lupus and lupus nephritis. It was the first FDA-approved biologic treatment within this indication. But according to Pharmacy Times, belimumab is now being looked at as a potential therapeutic option for patients living with systemic sclerosis. The U.S. Food and Drug Administration (FDA) recently granted Orphan Drug designation to belimumab for systemic sclerosis, or systemic scleroderma.
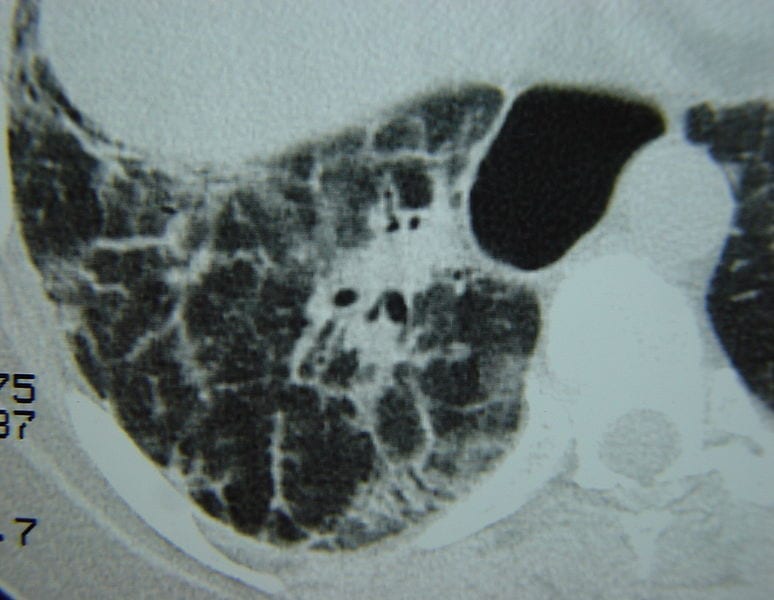
Many patients living with systemic scleroderma also have issues with lung disease. As a result, new research has been focused on relieving lung disease symptoms and improving outcomes within this patient population. Moving forward, GSK hopes to launch a Phase 2/3 study evaluating the treatment for patients within this community.
What is Orphan Drug Designation?
Orphan Drug designation is granted to drugs or biologics intended to treat, diagnose, or prevent rare conditions. These are defined as conditions affecting fewer than 200,000 Americans. This designation comes alongside incentives for drug developers, such as fee waivers, increased regulatory assistance, seven years of market exclusivity upon drug approval, and tax credits.
What is Belimumab?
DrugBank Online explains that belimumab is:
a fully human recombinant IgG1λ monoclonal antibody that inhibits soluble human B lymphocyte stimulator protein (BLyS, also referred to as BAFF and TNFSF13B), a B cell survival factor. By binding to BLyS and blocking its interaction with B cell receptors, belimumab inhibits the survival of B cells.
You can learn more about belimumab here.
An Overview of Systemic Sclerosis
Systemic sclerosis, also known as systemic scleroderma, is a rare autoimmune disorder characterized by scar tissue (fibrosis) in the skin and internal organs, which causes skin thickening and tightening. Systemic sclerosis affects both the skin and internal organs, while localized sclerosis affects only specific parts of the body. An estimated 40,000-165,000 people within the United States are living with systemic sclerosis. It is significantly more common in females than in males.
Symptoms, which typically manifest in middle age, can include:
- Hardened or tightened patches of skin
- Raynaud’s phenomenon (fingers and toes that turn blue or white in response to cold temperature)
- High blood pressure
- Shortness of breath
- Joint pain
- Diarrhea
- Intestinal pseudo-obstruction
- Open sores on the fingers or painful bumps under the skin
- Heartburn
- Kidney function problems
- Swollen fingers and hands
No cures exist for systemic sclerosis. Treatments are largely symptomatic. Treatment options include physical therapy, antibiotics, immune-suppressive drugs, blood pressure medications, fibrosis-combatting medications, and pain medication (among others).