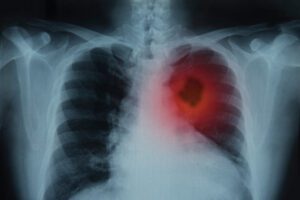
As reported on Business Wire, ImmunityBio has released new clinical findings suggesting that its interleukin‑15 superagonist, ANKTIVA® (nogapendekin alfa inbakicept), may help restore immune function and improve survival outcomes in patients with non-small cell lung cancer (NSCLC) when administered alongside established checkpoint inhibitor therapies. Results were derived from two clinical studies—QUILT‑2.023 and QUILT‑3.055—spanning 151 patients with disease ranging from treatment‑naïve to heavily pretreated.
Immune Restoration Observed Across Multiple Settings
In the first-line, randomized QUILT‑2.023 trial, patients receiving ANKTIVA combined with a checkpoint inhibitor experienced a statistically significant rise in absolute lymphocyte count (ALC) compared with those treated with checkpoint blockade alone. The improvement was not only significant (p=0.0065) but also sustained over time, supporting ANKTIVA’s role as an immune‑stimulating agent.
The QUILT‑3.055 trial, which enrolled patients who had already progressed following checkpoint therapy, showed similar trends. In this later‑line population, 77% achieved restoration or maintenance of an ALC ≥1.0 × 10³ cells/µL, meeting the study’s predefined threshold for immune response.
Higher Immune Competence Linked to Longer Survival
Immune recovery proved clinically meaningful. Responders in QUILT‑3.055 lived notably longer than non‑responders, with median overall survival of 16.2 months compared with 11.8 months (HR 0.52; p=0.0369).
Patients achieving stronger immune reconstitution—defined as reaching an ALC ≥1.2 × 10³ cells/µL—fared even better. This subgroup recorded a median overall survival of 21.1 months (HR 0.33; p=0.0009), regardless of PD‑L1 expression. These outcomes exceed the typical 7–9‑month median survival seen historically with standard second‑line chemotherapy such as docetaxel.
Challenging the Role of Chemotherapy in Later-Line NSCLC
Checkpoint inhibitors have revolutionized NSCLC treatment, yet most patients eventually relapse. According to ImmunityBio founder Patrick Soon‑Shiong, M.D., these results may support a shift away from cytotoxic chemotherapy in patients who fail initial immunotherapy.
He noted that ANKTIVA’s activation of both NK cells and CD8+ T cells may represent what the company calls “Immunotherapy 2.0”—a strategy aimed at reversing immune exhaustion rather than simply blocking inhibitory pathways.
Ongoing Phase 3 Trial to Confirm Findings
A randomized Phase 3 trial (ResQ201A) is now underway, comparing ANKTIVA plus checkpoint inhibition against docetaxel in second‑line NSCLC. The study is intended to confirm survival outcomes and potentially support regulatory submissions.
About the Studies
QUILT‑2.023 evaluated ANKTIVA alongside standard PD‑1/PD‑L1 therapies in untreated advanced NSCLC. Although enrollment closed early due to shifts in the first‑line treatment landscape, the trial provided key biologic data demonstrating ANKTIVA’s immune‑restoring effect.
QUILT‑3.055 enrolled patients with various solid tumors who had previously responded to but later progressed on checkpoint inhibitors. Participants continued their existing checkpoint therapy and received ANKTIVA in repeated cycles, with overall survival analyzed through the lens of immune recovery.
Looking Ahead
ImmunityBio plans to publish the full data set in a peer‑reviewed journal and present additional findings at future scientific meetings. The company also noted that ANKTIVA, when used with checkpoint inhibitors, is covered by multiple patents extending into the mid‑2030s.