As reported by Business Wire, Enanta Pharmaceuticals has initiated its first clinical study of EDP-978, a novel oral inhibitor of the KIT receptor tyrosine kinase being developed for chronic urticaria and other mast cell–mediated disorders. The company announced that the first participant has been dosed in a Phase 1 trial designed to assess the compound’s safety and early clinical profile in healthy volunteers.
The randomized, double-blind, placebo-controlled study represents the first time EDP-978 has been tested in humans. It will include both single-ascending dose and multiple-ascending dose phases, enrolling approximately 98 adults between the ages of 18 and 65. In addition to evaluating safety and tolerability, the trial will characterize pharmacokinetics and pharmacodynamic effects, including changes in serum tryptase, a biomarker associated with mast cell activity. A dedicated cohort will assess the impact of food on drug exposure, and the multiple-dose phase will involve 14 days of once-daily dosing. Initial results are expected in the fourth quarter of 2026.
EDP-978 is Enanta’s lead immunology program and is designed as a once-daily oral therapy. Preclinical testing has shown the molecule to be highly potent and selective for KIT, with strong activity in binding and cellular assays and robust in vivo efficacy at very low concentrations. The compound has also demonstrated favorable absorption, distribution, metabolism, and excretion characteristics in preclinical models, supporting its advancement into clinical development.
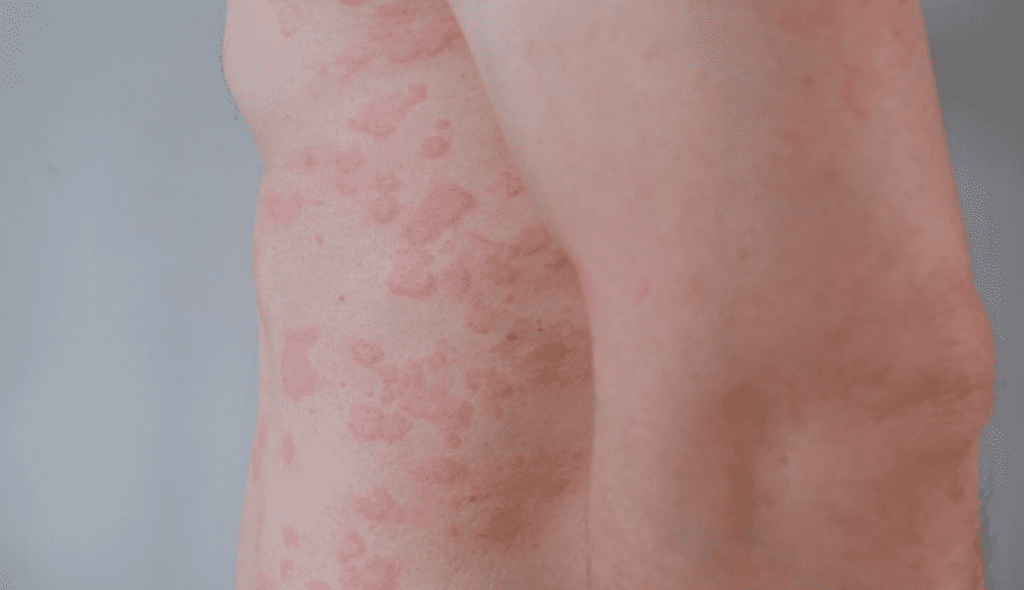
Chronic urticaria is a persistent inflammatory skin condition driven by mast cell activation and characterized by recurrent hives, angioedema, or both lasting longer than six weeks. The disease encompasses chronic spontaneous urticaria, which lacks a consistent trigger, as well as chronic inducible urticaria, in which symptoms are reliably provoked by stimuli such as pressure, cold, heat, or sunlight. Both forms can significantly disrupt daily life due to intense itching, visible skin lesions, sleep impairment, and associated psychological distress.
Despite available treatments, a meaningful proportion of patients remain inadequately controlled, highlighting the need for new therapeutic options, particularly oral agents that can be taken once daily. By targeting KIT, a key regulator of mast cell survival and activation, Enanta aims to address the underlying biology of the disease rather than only its symptoms.
If successful, the Phase 1 results will inform further clinical development of EDP-978 and help determine its potential role as a new treatment option for patients with chronic urticaria and related mast cell–driven conditions.