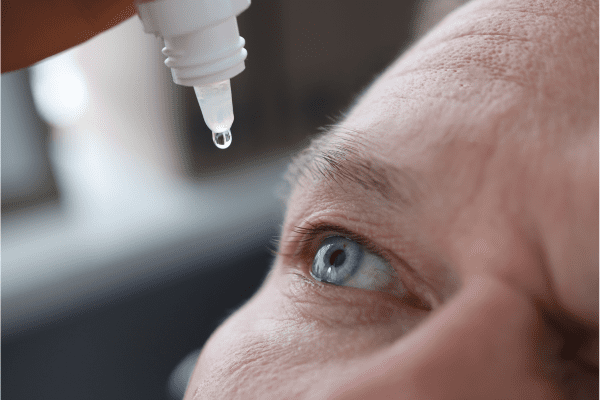
Health Canada has officially approved Cysklar® (cysteamine ophthalmic solution) 0.44%, marking a significant breakthrough for Canadians living with cystinosis. According to BusinessWire.com, the approval enables patients to access a topical eye drop treatment specifically designed to address corneal cystine crystal accumulation, a hallmark complication of this rare genetic disorder.
Understanding the Challenge
Cystinosis is a rare but serious multi-system genetic disorder characterized by the harmful accumulation of cystine crystals throughout the body. These crystals don’t limit themselves to one organ—they can accumulate in the eyes, kidneys, liver, muscles, and other vital systems. When cystine crystals deposit in the cornea of the eye, they can cause significant vision problems and discomfort for patients, making effective treatment essential for maintaining quality of life.
The Solution
Cysklar represents a major therapeutic advancement for managing corneal complications in cystinosis patients. As a topical eye drop solution, it offers a straightforward and patient-friendly approach to treating corneal cystine crystal accumulation. The ease of use, simply applying drops to the affected eyes, makes this treatment accessible and practical for patients of all ages, including children who may require parental administration.
A Commitment Spanning Two Decades
The approval reflects Leadiant Biosciences’ unwavering dedication to the cystinosis community. The pharmaceutical company has invested more than 20 years in serving this population, and the launch of Cysklar demonstrates their ongoing commitment to providing effective therapeutic options. According to Giuseppe Izzi, CEO of Leadiant Biosciences, this milestone showcases the company’s promise to continue developing care solutions for people living with cystinosis.
Significance for the Canadian Cystinosis Community
For Canadian patients and families affected by cystinosis, this approval represents hope and tangible progress. Treatment options specifically targeting corneal complications have been limited, and the availability of Cysklar provides a new tool in managing this multifaceted disease. By offering an easy-to-use topical treatment, the approval helps reduce the burden on patients and caregivers who are already managing complex medical needs.
Moving Forward
Leadiant Biosciences is committed to keeping patients, caregivers, and healthcare providers informed about Cysklar’s availability in Canada. The company plans to communicate important details about timing and distribution, ensuring that those who could benefit from this treatment have clear information about access options.