
Dark Sweet Cherries Against Breast Cancer
Pictured above: Giuliana Noratto, Ph.D., (left) and Lauren Stranahan, Ph.D., led research showing that compounds in dark sweet cherries may help slow the growth and

Pictured above: Giuliana Noratto, Ph.D., (left) and Lauren Stranahan, Ph.D., led research showing that compounds in dark sweet cherries may help slow the growth and
Daiichi Sankyo has taken a significant step toward expanding treatment options in Japan by submitting regulatory approval for Enhertu, an advanced cancer-fighting drug designed to

As reported by FiercePharma, the U.S. Food and Drug Administration has approved the first fully independent generic version of GlaxoSmithKline’s widely used asthma inhaler, Flovent,

You can not see, hear, or touch my rare condition. I STINK! (LITERALLY) I wasn’t sure if I was going to tell my story. But

Researchers have created a fundamentally new class of HIV-fighting molecules by combining two separate antiviral mechanisms into a single engineered protein. Early laboratory results, reported

As reported on BioPharmaDive, UCB has reported positive results from a head-to-head clinical trial showing that its anti-inflammatory biologic Bimzelx was more effective than AbbVie’s

Editor’s Note: Patient Worthy is proud and honored to share Jennifer’s story with cystic fibrosis and colon cancer, originally published on the Cystic Fibrosis Research

The biotechnology sector experienced significant positive developments this week, with Xenon Pharmaceuticals’ seizure medication achieving a decisive late-stage victory while regulatory pathways accelerated for several
As reported on Healio, glucagon-like peptide-1 (GLP-1) receptor agonists, best known for treating diabetes and obesity, are emerging as unexpected players in breast cancer care.

CAR T- cells are immune cells that are engineered in a laboratory to attack cancer. The therapy has worked as planned for certain blood cancers

Artios Pharma has achieved a significant milestone in cancer treatment development with the U.S. Food and Drug Administration’s granting of Fast Track designation to ART6043,

As reported on drugs.com, Bristol Myers Squibb (BMS) has announced encouraging interim results from the Phase 3 portion of the SUCCESSOR-2 clinical trial evaluating oral

As reported on FierceBioTech, the U.S. Food and Drug Administration has released its complete response letter (CRL) detailing why it declined to approve Regenxbio’s experimental

As covered on PharmaBiz, Roche has reported topline results from the phase III persevERA Breast Cancer study, which evaluated the oral selective oestrogen receptor degrader

Editor’s Note: Patient Worthy is proud to share this article from our friends at the Courageous Parents Network. A parent often can sense when there

Daiichi Sankyo has taken a significant step forward in expanding treatment options for early-stage breast cancer patients by submitting a supplemental New Drug Application (sNDA)

As reported in a recent article by Pharmaceutical Technology, Bristol Myers Squibb (BMS) has secured US Food and Drug Administration (FDA) approval for Sotyktu (deucravacitinib)
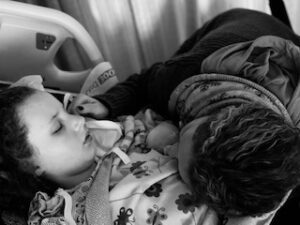
Editor’s Note: Patient Worthy is honored to share this caregiver story with you, originally written by Aisling Melton, Parent Champion at Courageous Parents Network. In
We're honored to share Heather's story as a caregiver for her husband Nathan, who is living with Huntington's disease. Read here:
patientworthy.com/2026/03/28/heather-and-nathans-journey-with-huntingtons-disease/
#PatientWorthy #shareyourstory #PatientStory #HD #huntingtons #HuntingtonsDisease
... See MoreSee Less

Colorectal Cancer Awareness Month: Colorectal cancer is preventable, treatable, and survivable—especially when caught early. Swipe through these 5 important facts about prevention, screening, and advances in treatment. #ColorectalCancerAwarenessMonth #colorectalcancer #FactFriday #PatientWorthy
#shareyourstory with us here: bit.ly/4dV7gru
... See MoreSee Less


 +4
+4
Understanding Testing for Non-Small Cell Lung Cancer with HER2 Mutation- ✨
When diagnosed with NSCLC, testing for genetic mutations like HER2 is key too choosing the right treatment. From biopsies to advanced tests like NGS & FISH, identifying the HER2 mutation helps doctors target therapies that can make a difference. #LungCancerAwareness #HER2Mutation #PatientWorthy
... See MoreSee Less


© Copyright 2024 Patient Worthy

Sign Up With a Patient Worthy Account and Share Your Rare Story
- OR -

Make a difference, share your experiences and get paid. Opt-in and join Patient Worthy's panel for paid opportunities such as surveys, market research, patient advisory panels and more.
What best describes you when it comes to rare disease? (check all that apply)

- OR -

Make a difference, share your experiences and get paid. Opt-in and join Patient Worthy's panel for paid opportunities such as surveys, market research, patient advisory panels and more.