Age: 43, Body: 80. When Ageing Moves at a Different Pace.
For most of us, our body follows a predictable timeline when it comes to growing older. While there's some variation depending on genes and lifestyle, each year, we age with…
For most of us, our body follows a predictable timeline when it comes to growing older. While there's some variation depending on genes and lifestyle, each year, we age with…

According to a press release from Alnylam Pharmaceuticals, the Massachusetts-based biopharmaceutical company has completed the submission of its New Drug Application (NDA) for givosiran — its experimental acute hepatic porphyria…
Functional neurological disorder is a conversion disorder. This type of complex disorder is often thought to be linked with psychological symptoms, serious stress, or mental distress. With functional neurological…
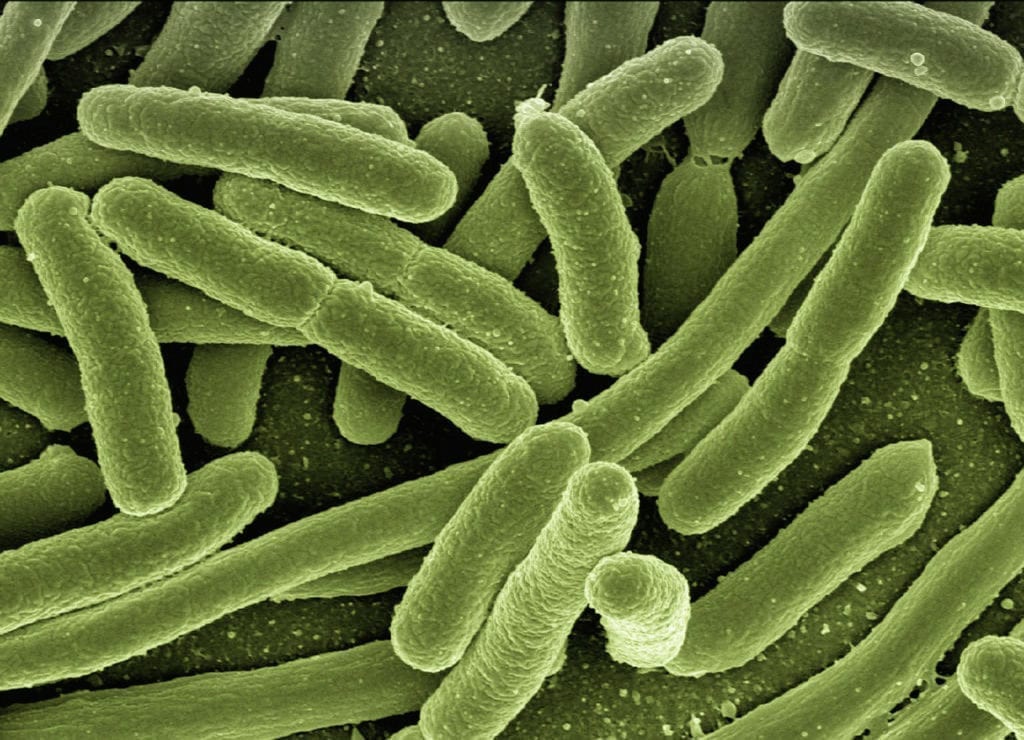
According to a story from bioengineer.org, a team of researchers associated with Oregon State University are making further headway in understanding the connection between our health and the bacteria that…
Stem Cell Therapies While stem cell therapies hold a great deal of promise for many rare diseases, there is still a lot that we don't understand. Experimental stem cell therapies…
Meredith Resnick's breast cancer risk is 42%. This risk is made up of her higher breast density (denser breasts are associated with increased risk), Ashkenazi Jewish heritage, a family history…

Do you have ADPKD? If you or someone you know has ADPKD, email us at [email protected]. We'd love to hear from you about your experience! According to a story from…
In the mid-1980s the FDA was criticized for being slow in approving new drugs. In fact, a commission was formed as a result of the mounting criticism. Janet Woodcock,…

According to a story from ANCA Vasculitis News, a recent study conducted in Korea determined that ANCA vasculitis patients in the region were at a greater risk of developing thyroid…
"A new way to improve the efficacy of a drug taken by millions of patients throughout the world." Background Adalimumab (also known as Humira) and infliximab (also known as Remicade)…