NXP800 for Cholangiocarcinoma Earns Orphan Drug Designation
In the United States, the FDA grants Orphan Drug designation to drugs or biologics that are intended to treat, diagnose, or prevent rare conditions. A rare condition is one…
In the United States, the FDA grants Orphan Drug designation to drugs or biologics that are intended to treat, diagnose, or prevent rare conditions. A rare condition is one…

245 words (source - 6% match) vs. 386 words (mine - 4% match) In the European Union, Orphan designation is granted to therapies intended to treat or use for a…

Previously approved for the treatment of narcolepsy, pitolisant (marketed under the brand name Wakix) may soon be leveraged for another rare condition: idiopathic hypersomnia (IH). Pitolisant is a histamine 3…
INT230-6, developed by clinical-stage biotechnology company Intensity Therapeutics (“Intensity”), is an investigational therapy comprising three moieties: cisplatin, SHAO-FA (a penetration enhancing molecule), and vinblastine sulfate. A moiety, in this sense,…
At the moment, there are limited treatment options for glioblastoma, a rare form of brain cancer. While surgery, radiation, chemotherapy, and electric-field therapy are all utilized in the fight…

Despite advances in research and the advent of multiple investigational treatments, pancreatic cancer is still incredibly difficult to treat. The five-year survival rate sits at just 12%. Existing therapies…
NS Pharma, Inc., a subsidiary of Nippon Shinyaku Co., recently announced via news release that its therapy NS-089/NCNP-02 was granted Orphan Drug designation from the U.S. Food and Drug…
Even with available therapies, pancreatic cancer is incredibly tough to treat and comes with a poor prognosis. Only 12% of people diagnosed with pancreatic cancer are still alive after…
Orphan drug designation was first introduced to the European Union in 2000. The designation is granted to therapies intended to treat, diagnose, or prevent a rare, life-threatening, or chronically…
Have you ever heard of Orphan Drug designation? This special status was created to incentivize the development of therapies for people with rare conditions (affecting fewer than 200,000 people…
Earlier this year, the FDA granted Accelerated Approval to ELEVIDYS (delandistrogene moxeparvovec-rokl) for the treatmnt of Duchenne muscular dystrophy (DMD) in boys aged 4-5. While this is a stunning…
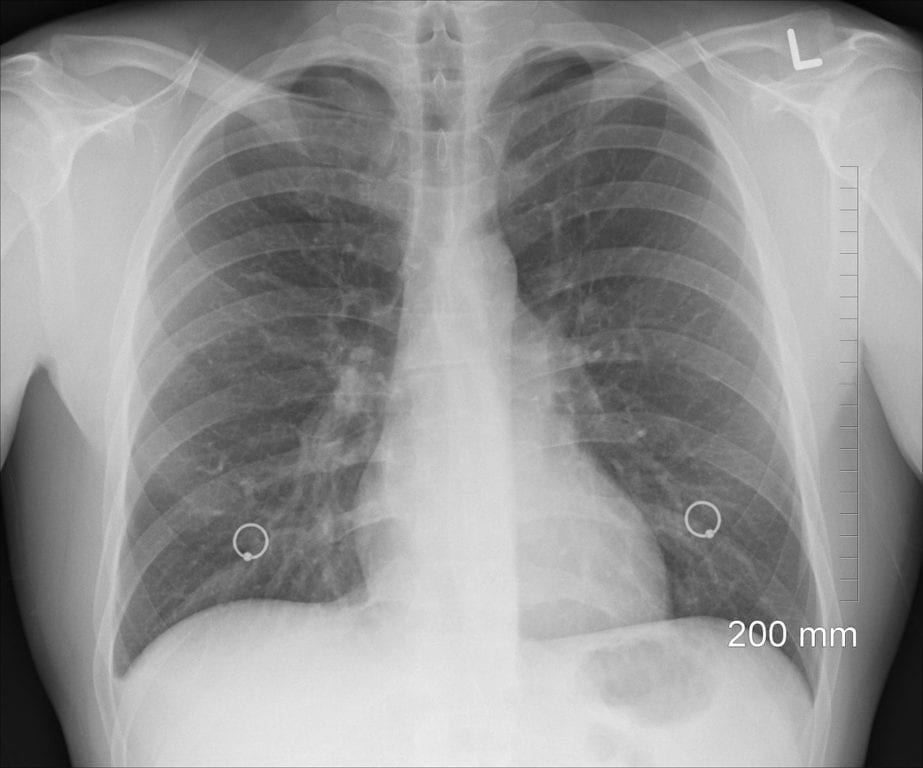
According to an article from Akosua Mireku of Pharmaceutical Technology, the U.S. Food and Drug Administration recently granted Orphan Drug designation to SNB-101, a potential therapy for people living with…
Orphan Drug designation is granted to therapies that are designed to treat, diagnose, or prevent rare diseases or conditions. In the United States, a rare condition is one affecting fewer…
Medical research has yielded a significant number of breakthroughs that have advanced the field and helped people in need. But in the rare disease sphere, it has sometimes been…
Throughout the world, it can be difficult to advance research and drug development in the rare disease space. In part, this is because many rare diseases have small population…
In May 2023, Pharmaceutical Technology reported that INO-3107, a DNA medicine vaccine candidate developed by biotechnology company Inovio Pharmaceuticals (“Inovio”), received Orphan Drug designation from the European Commission. INO-3107…
Gene therapy has the potential to improve outcomes among individuals across various disease spectrums. In the case of clinical-stage biotechnology company Rocket Pharmaceuticals, the company is working to develop…
Mesothelioma can sometimes be difficult to treat. The cancer can spread to nearby tissue and nerves, and often isn’t diagnosed until later stages. Admittedly, there are a number of…

Although the 5-year survival rate for pancreatic cancer has slightly risen (from 11% to 12%), there is still much work to be done. Pancreatic cancer can be difficult to treat.…
In mid-May 2023, Pharmaceutical Technology reported that a new combination treatment had earned Orphan Drug designation. This combo? Obeticholic acid (OCA) and bezafibrate. Together, they represent a potential therapeutic…
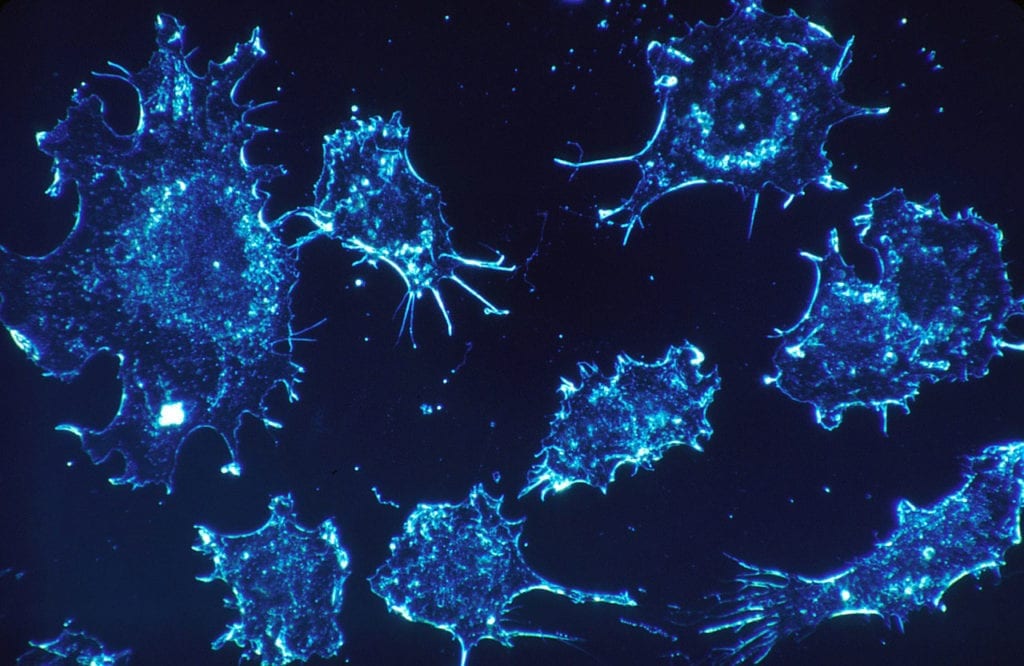
Pyxis Oncology is driven to improve the lives of people living with cancer through the development of innovative immunotherapy and antibody drug conjugate (ADJ) therapies. Their treatment PYX-201, a novel…
The passage of the Orphan Drug Act was an important step in creating an environment that supports rare disease research and drug development. This Act created Orphan Drug designation,…
In February 2023, I spoke with Dr. Allen Davidoff of XORTX Therapeutics Inc. (“XORTX”) about the company’s commitment to developing therapies for people living with progressive kidney disease. We…
Prior to the end of March 2023, ganaxolone had received six separate Orphan Drug designations from the FDA; more recently, as shared in a news release from commercial-stage pharmaceutical…

Cases of multiple myeloma seem to be rising across the country and the globe. New and effective therapeutic interventions have the potential to significantly benefit this patient population. Ichnos Sciences…