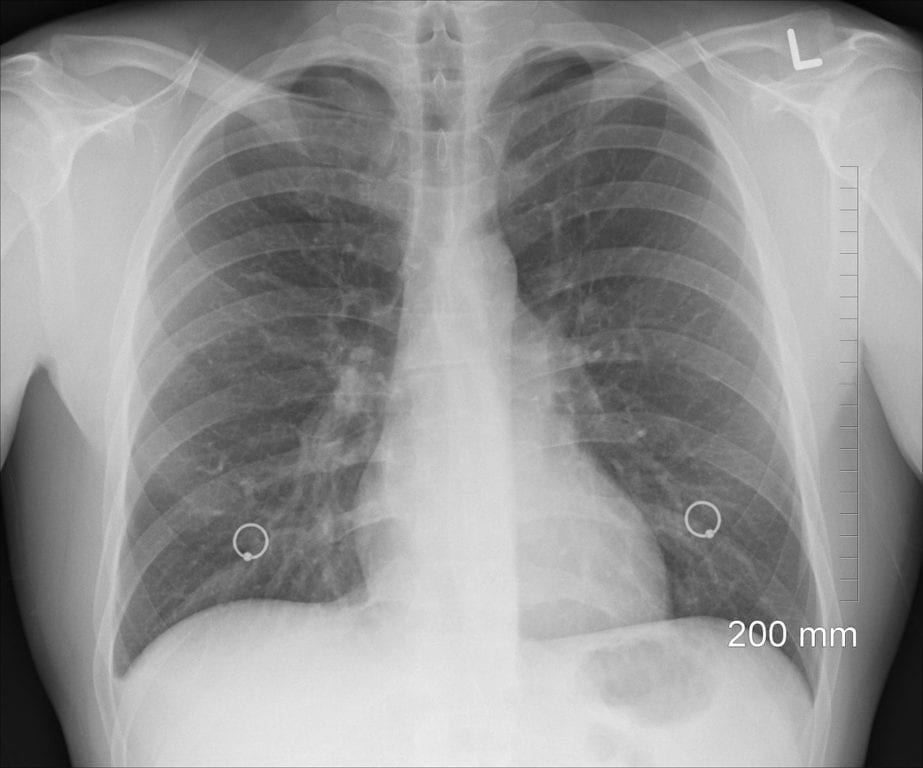
Taldefgrobep Alfa Earns EU Orphan Drug Status for Spinal Muscular Atrophy (SMA)
Orphan drug designation was first introduced to the European Union in 2000. The designation is granted to therapies intended to treat, diagnose, or prevent a rare, life-threatening, or chronically…