Boehringer Ingelheim’s More Than Scleroderma Educational Webinar
More Than Scleroderma: A Live Educational Event August 19, 2020 at 5:30 PM ET Join us for a webinar about scleroderma Because scleroderma is so rare, it can be hard…
More Than Scleroderma: A Live Educational Event August 19, 2020 at 5:30 PM ET Join us for a webinar about scleroderma Because scleroderma is so rare, it can be hard…
More Than Scleroderma: A Live Educational Event August 13, 2020 at 2:00 PM ET Join us for a webinar about scleroderma Because scleroderma is so rare, it can be hard…
Thanks to the rampant spread of COVID-19, many events, symposiums, and clinics moved online. One was the 2020 ACR State-of-the-Art Clinical Symposium, in which participants discuss diagnostic information, treatments,…
Biotechnology company Talaris Therapeutics, Inc. submitted an Investigational New Drug (IND) application to the FDA for their novel cell therapy treatment FCR001. As per a press release, the FDA…
According to a story from Scleroderma News, June is recognized as Scleroderma Awareness Month. While physical distancing as a result of the ongoing coronavirus/COVID-19 pandemic may keep some folks at…
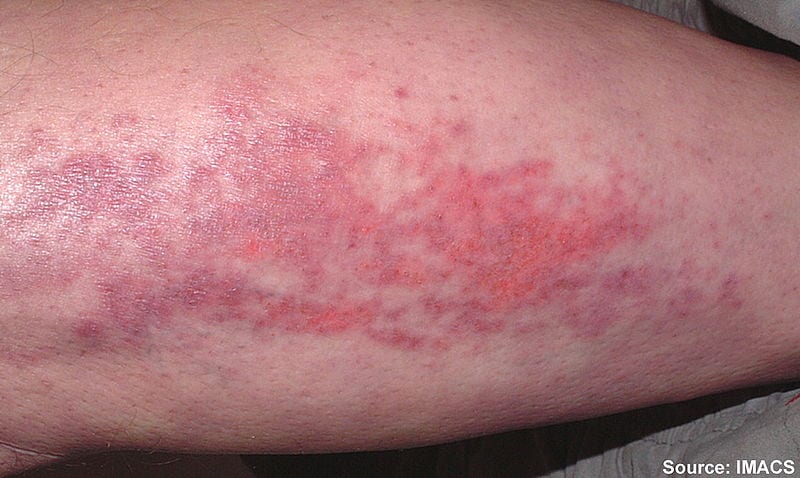
This year, because of COVID-19, many healthcare-related events and symposiums have been moved online. However, that doesn't mean that the insights are any less helpful! In the virtual ACR…
by Danielle Bradshaw from In The Cloud Copy A year-long study has shown that Tracleer greatly reduces the skin-related symptoms of scleroderma. The results also showed that although it helped…
CSL Behring has recently received the Orphan Drug designation for Privigen for the treatment of systemic scleroderma. Privigen has also been granted the Fast Track designation. This therapy has been…
By Jodee Redmond from In The Cloud Copy A recent study suggests that certain antibodies could be linked to specific human leukocyte antigen (HLA) genes and environmental components in scleroderma.…
According to a story from Benzinga, the biotherapeutics company CSL Behring has recently announced that its medical product PRIVIGEN ® has earned Orphan Drug designation from the US Food and…
According to a story from Pulmonary Hypertension News, a recent study has determined that cardiopulmonary exercise testing can be an effective method to help identify systemic sclerosis patients that are…
It is already known that gender plays a role in systemic sclerosis disease with scleroderma – it’s about four times as prevalent in women than men - but a…

According to a story from Scleroderma News, the results of a recent mouse study could have significant implications for the future of scleroderma treatment. In the study, scientists used a…

According to a story from American Nurse Today, the US Food and Drug Administration (FDA) approved a treatment on September 6th, 2019 for interstitial lung disease associated with scleroderma, a…
According to a story from The Rheumatologist, evidence suggests that patients with rare diseases, such as autoimmune diseases, can benefit from visiting combined clinics, which are special medical centers in…

According to a story from finanzen.at, the drug nintedanib (marketed as Ofev) has officially been approved by the US Food and Drug Administration (FDA) as a treatment for interstitial lung…
According to a story from Financial Buzz, the drug developer Emerald Health Pharmaceuticals, Inc., recently announced that the company had completed a phase 1 clinical trial for its experimental drug…

According to a story from Comic Sands, mother Alison Beesley is now committed to taking care of her daughters Eliza and Eleanor full time. Eliza is nine years old and…
According to a story from Scleroderma News, a recent study has recommended that scleroderma patients with a rare, poorly understood form of interstitial lung disease (ILD) should receive regular monitoring…
According to a story from Medpage Today, a recent study testing the medication abatacept (marketed as Orencia) as a treatment for diffuse cutaneous systemic sclerosis concluded with rather mixed results.…
According to a publication from Scleroderma News, a recent study suggests that blood concentrations of certain immune cells could correlate with the severity of cases of systemic sclerosis. About Systemic…

According to a story from Healio, the US Food and Drug Administration's (FDA) Arthritis Advisory Committee recently voted 10-7 in favor of recommending the drug nintedanib as a treatment for…
An article from Comic Sands recently covered the story of a UK mother, Alison Beesley, who is now committed to taking care of her daughters Eliza and Eleanor full time. Eliza is nine years old and…

According to a story from Scleroderma News, a recent study has identified a host of risk factors that can increase a patient's likelihood of experiencing progressive organ damage. The identification…
According to a story from Scleroderma News, a team of researchers associated with Michigan Medicine has been awarded a grant to the tune of $10.2 million from the National Institutes…