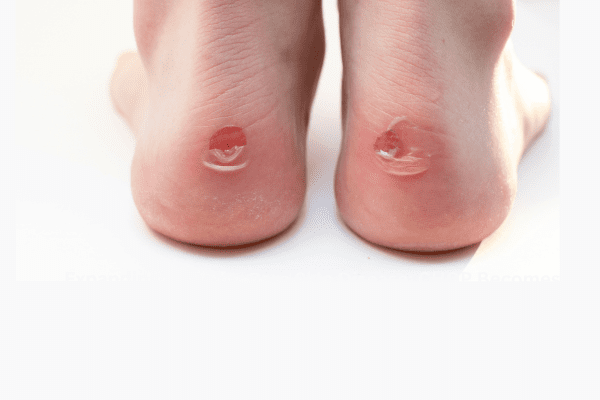
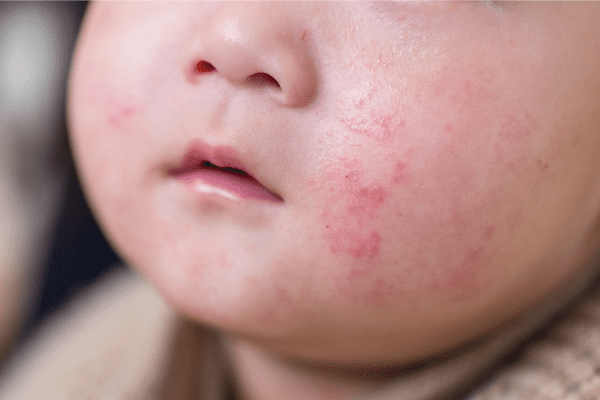
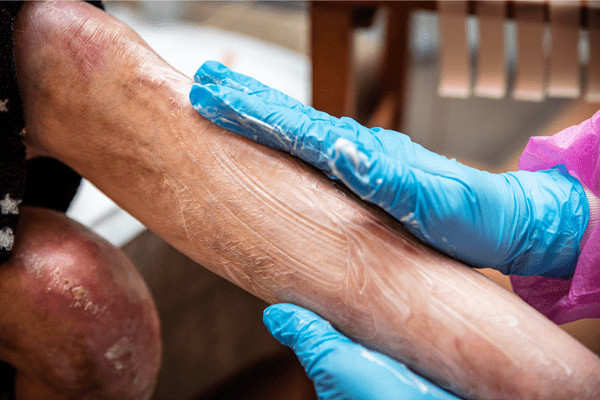
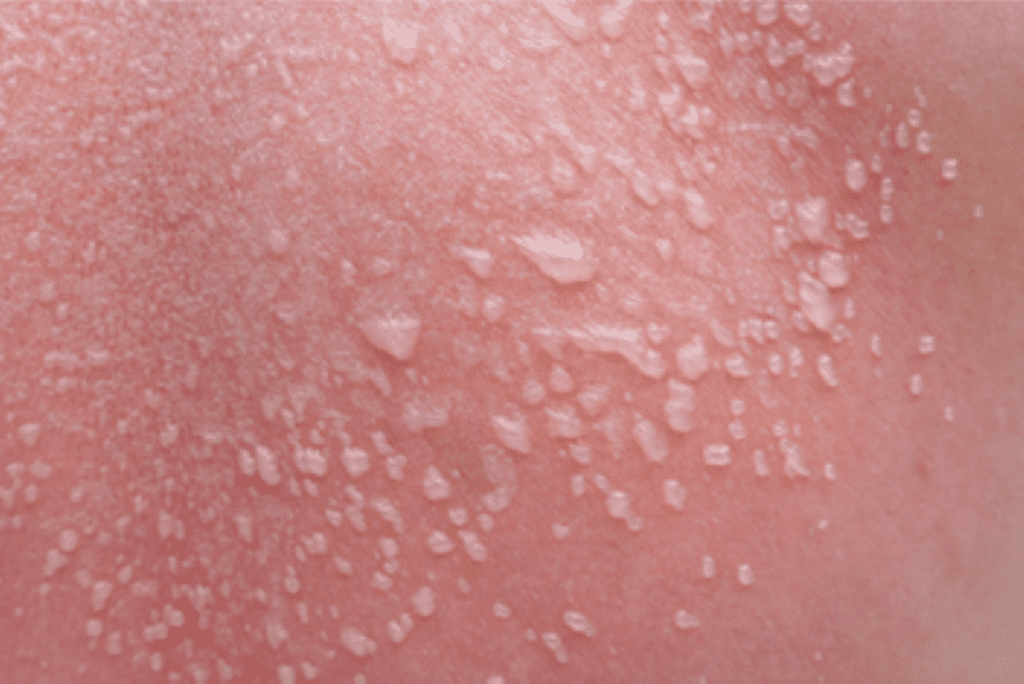
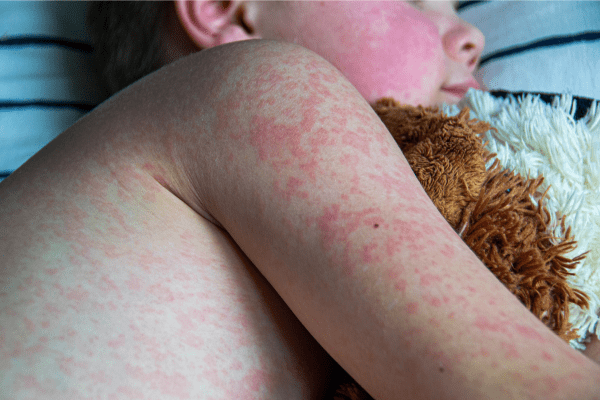
Epidermolysis Bullosa Care Enters a New Era With Gene-Targeted and Wound-Healing Therapies
An article from Medscape highlighted recent advances in treatment that are reshaping the clinical approach to epidermolysis bullosa (EB), transitioning care from largely supportive management to therapies that address genetic…