Pruitt’s Legacy: Raising Awareness of OTC Deficiency and Genetic Testing
Rare Disease Month is deeply personal to our family. Before Pruitt, our youngest son, we had never heard of Ornithine Transcarbamylase (OTC) Deficiency, nor did we truly understand how fragile…


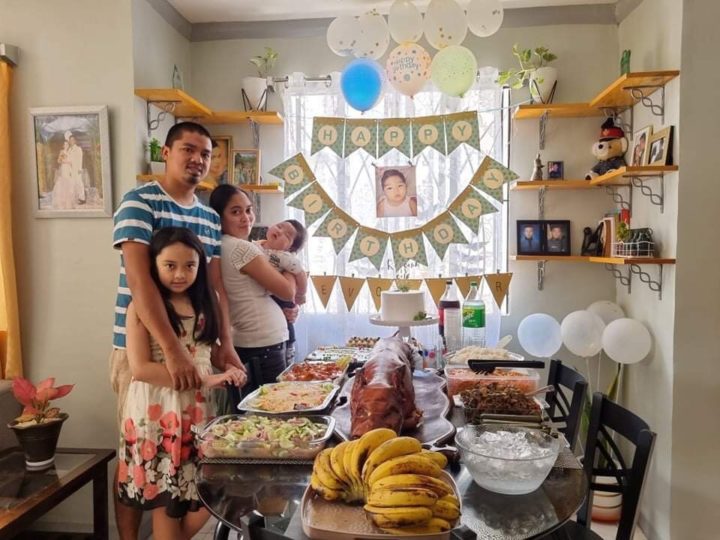


.jpg)



