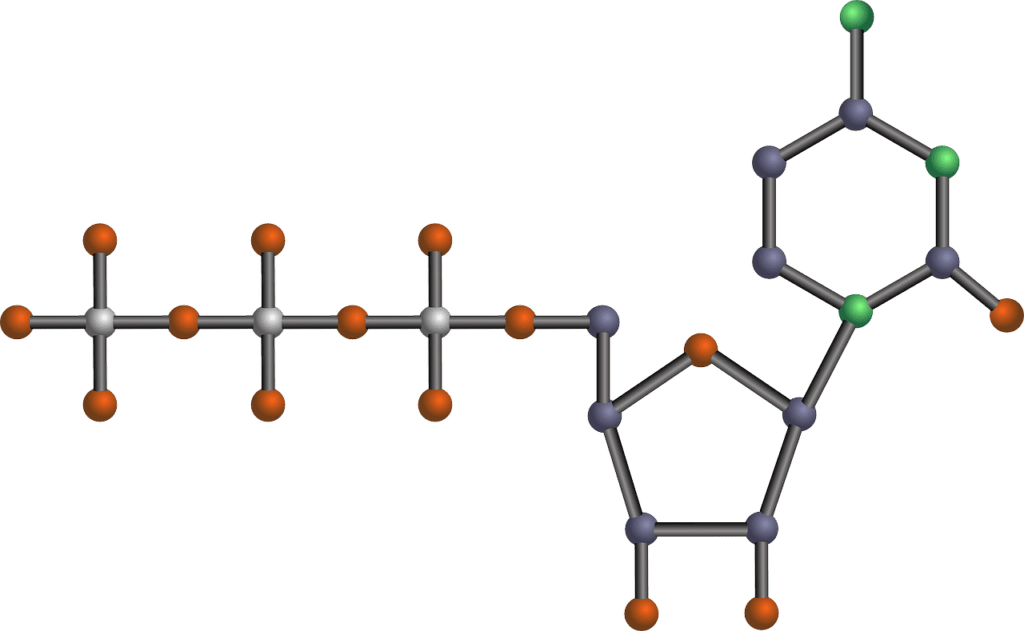
The Patriots Raise Awareness for Sickle Cell Anemia and Duchenne Muscular Dystrophy
This past Sunday, December 2nd, during the Patriot's game against the Minnesota Vikings, 20 players wore customized cleats to spread awareness for various philanthropic causes. This is the third year…