It Took Almost 60 Years for the Correct Diagnosis and Treatment for Primary Immunodeficiency
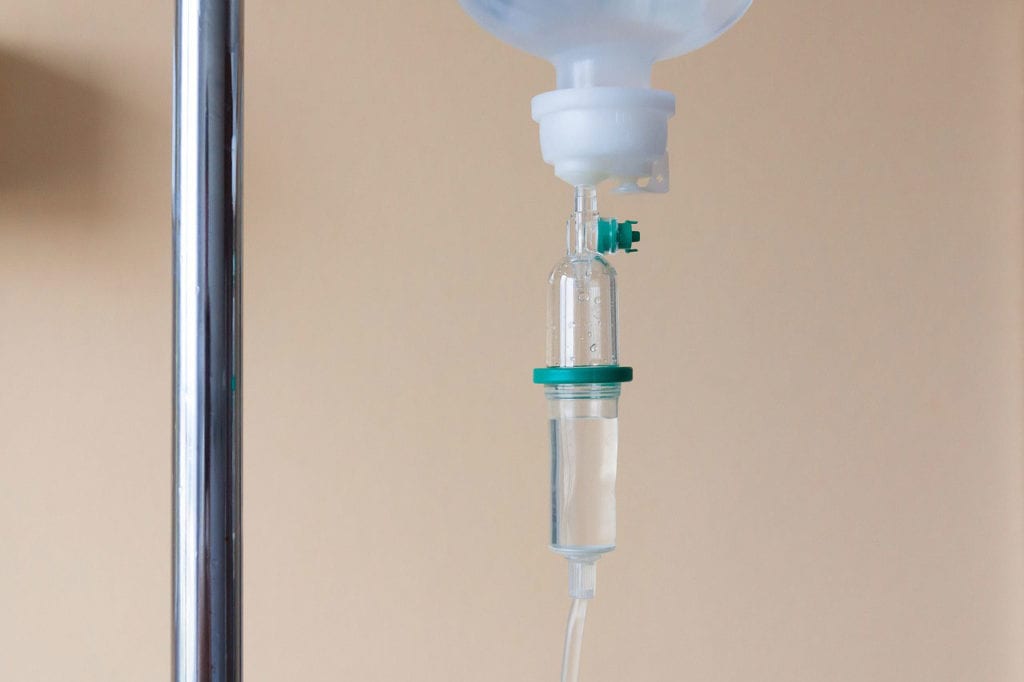
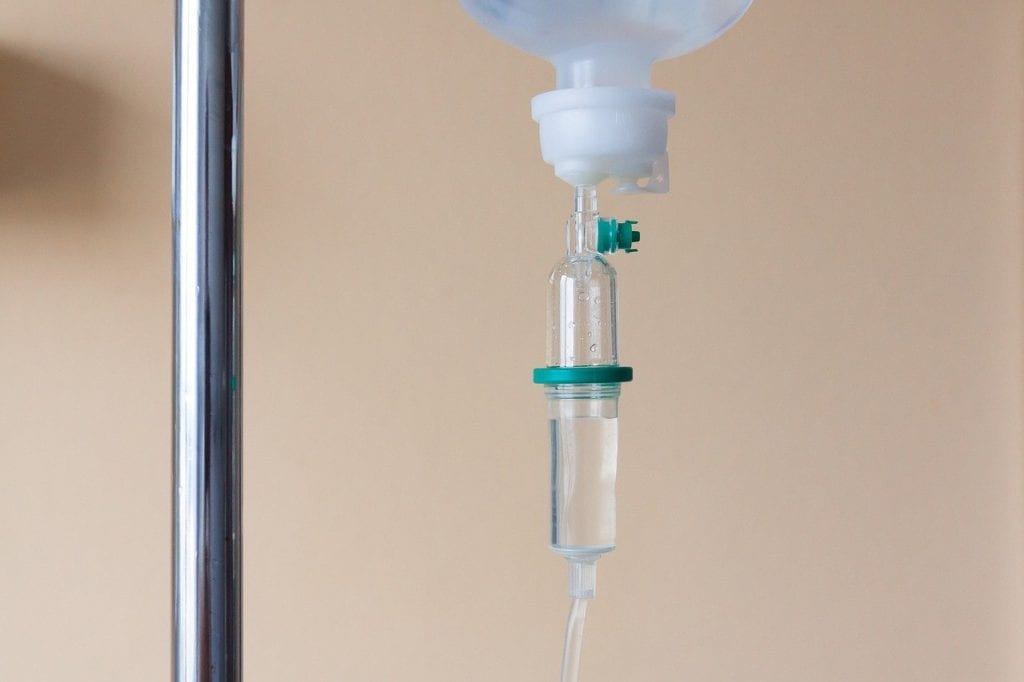
Eighty-five-year-old Frank Meuers considers his 500th immunoglobulin infusion to be a cause for celebration. Frank has logged details of his doctor’s visits since his first dose of replacement therapy with…