
Ensartinib Approved in China for ALK+ NSCLC as 1st Line Treatment
Two years ago, China approved ensartinib as a second-line therapy for those with ALK+ non-small cell lung cancer (NSCLC). There have been advances over time. Now, according to a late…

Two years ago, China approved ensartinib as a second-line therapy for those with ALK+ non-small cell lung cancer (NSCLC). There have been advances over time. Now, according to a late…
In a Phase 3 clinical trial, researchers evaluated I/ONTAK (also known as E7777) as a potential therapeutic option for persistent or recurrent cutaneous T-cell lymphoma (CTCL). According to Targeted Oncology,…
In a late March 2022 news release from biopharmaceutical company CANbridge Pharmaceuticals, Inc. ("CANbridge"), the company noted that the first patient was dosed in a Phase 1b/2 clinical trial evaluating…

Earlier this year, 4D Pharma Plc. ("4D Pharma") experienced a "win" when the FDA approved the company's plans to initiate a Phase 1 clinical trial evaluating MRx0005 and MRx0029 for Parkinson's…
On March 29, 2022, globally diversified chemical company LG Chem announced via press release that its Investigational New Drug (IND) application was approved by the U.S. Food and Drug Administration…
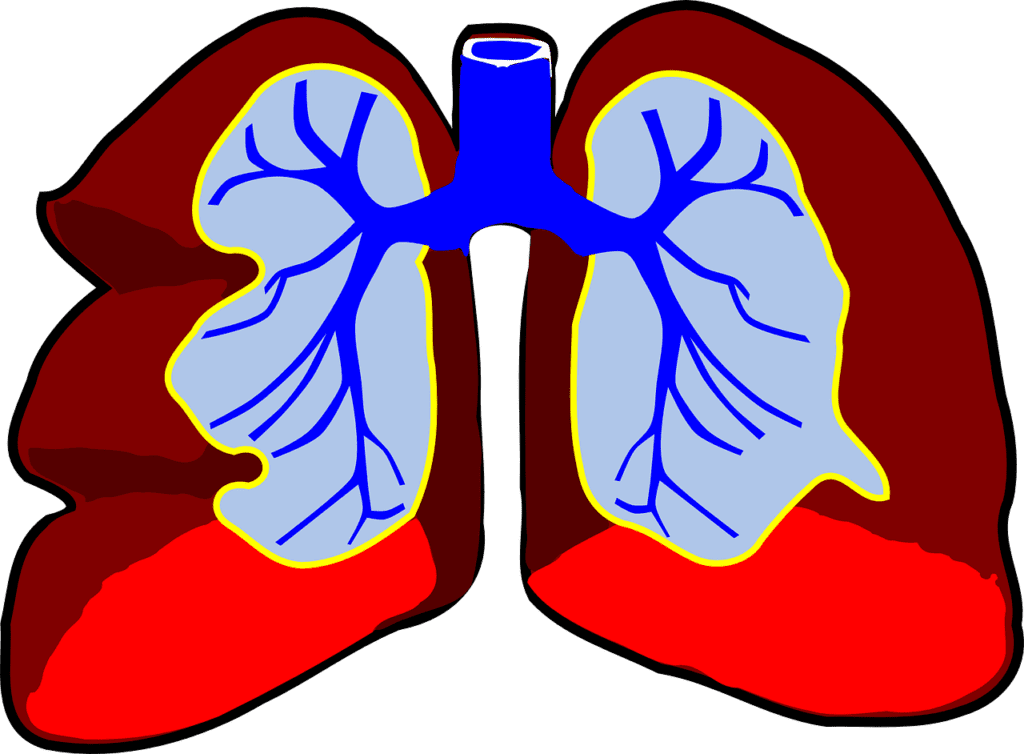
Currently, there are no cures for idiopathic pulmonary fibrosis (IPF), and the two available treatments aim to reduce lung scarring and symptom progression. However, it is clear that new therapeutic…

Without treatment, an estimated 20% of those with progressive keratoconus may eventually require corneal transplantation. Glaukos, a company creating novel therapies for chronic eye diseases, developed the iLink corneal cross-linking procedure to…
The American Academy of Allergy, Asthma & Immunology (AAAAI) Annual Meeting took place this year from February 25-28, 2022. During the meeting, stakeholders gathered to discuss new technologies, healthcare reform,…
According to a story from Targeted Oncology, the company Genmab has recently announced that the US Food and Drug Administration (FDA) has granted Orphan Drug designation to its investigational therapy…
ProQR Therapeutics is working to develop unique therapeutic solutions for genetic eye diseases. Currently, the company is evaluating its therapeutic candidate ultevursen within the Phase 2/3 Sirius and Celeste clinical trials; you can read…
Each year, an estimated 11,000 people across the globe are diagnosed with relapsed or refractory (R/R) T-cell acute lymphoblastic leukemia (T-ALL) or lymphoblastic lymphoma (LBL). However, there is only one…
Over the last few years, there has been much discussion around VLA1553, a Chikungunya virus vaccine candidate. The vaccine has so far earned Fast Track and Breakthrough Therapy designations within…
Clinical trials are greatly beneficial in understanding more about therapeutics and various conditions. For example, the Phase 3 VIITAL trial will evaluate the safety, efficacy, and tolerability of EB-101, an…
The first patient has been dosed in the Phase 1/2 B-LIEVE trial of FLT180a, an investigational treatment for hemophilia B. Freeline Therapeutics, the drug developer, announced the news in a…
In late February 2022, biopharmaceutical company Trascenta Holding Limited ("Trascenta") provided an update on its clinical program for TST001, a monoclonal antibody therapy designed for those with locally advanced or…
Cancer Network recently featured a press release by CTI BioParma announcing that the FDA has given accelerated approval for pacritinib to treat patients diagnosed with myelofibrosis and severely low platelets…

Karyopharm is currently developing selinexor, an investigational treatment for recurrent or advanced endometrial cancer. They had planned to submit a supplemental New Drug Application (sNDA) to the FDA, but now…
AstraZeneca and Daiichi-Sankyo are participating in a global collaboration to develop Enhertu (trastuzumab deruxtecan) for the treatment of HER2-low metastatic breast cancer (cancer that has spread). Enhertu is an ADC…
How effective is UPLIZNA on treating attacks associated with neuromyelitis optica spectrum disorder (NMOSD)? According to a news release from biopharmaceutical company Horizon Therapeutics plc ("Horizon"), an analysis highlighted that…
BRUKINSA (zanubrutinib) is a prescription medication which can be used to treat Waldenstrom macroglobulinemia (WM), mantle cell lymphoma, and marginal cell lymphoma. So far, the treatment has been approved in…
In a first-in-human, dose-escalation Phase 1 clinical trial, researchers are evaluating cosibelimab (CK-301) as a potential therapeutic option for those with metastatic cutaneous squamous cell carcinoma (cSCC). So far, shares Cancer…
Prior to February 2022, there were no approved disease-modifying therapies for those with pyruvate kinase (PK) deficiency. However, this has recently changed! According to a news release from biopharmaceutical company…

On February 16, 2022, biotechnology company Hillstream BioPharma Inc. (“Hillstream”) shared that its drug candidate HSB-1216 received Orphan Drug designation from the U.S. Food and Drug Administration. This particular designation…
On February 15, 2022, biotechnology company Zealand Pharma A/S (“Zealand”) announced that its Phase 3 1703 clinical trial evaluating dasiglucagon for congenital hyperinsulinism (CHI) had reached full enrollment. Within this…
In late January 2022, biopharmaceutical company BridgeBio Pharma, Inc. ("BridgeBio") shared via news release that the first patient had been dosed in the Phase 1/2 ADventure clinical trial. Within this…